Advertisements
Advertisements
प्रश्न
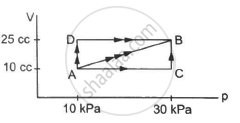
Figure shows three paths through which a gas can be taken from the state A to the state B. Calculate the work done by the gas in each of the three paths.
Advertisements
उत्तर
Work done during any process, W = P ∆ V
If both pressure and volume are changing during a process, then work done can be found out by finding the area under the PV diagram.
In path ACB, for line AC :-
Since initial volume is equal to final volume,
∆ V = 0
⇒ WAC = P ∆ V = 0
For line BC :-
P = 30 × 103 pa
WACB = WAC + WBC = 0 + P∆V
= 30 × 103 × (25 − 10) × 10−6
= 0.45 J
For path AB:-
Since both pressure and volume are changing, we use the mean pressure to find the work done.
Mean pressure, P = \[\frac{1}{2} \times (30 + 10) \times {10}^3\]
WAB = \[\frac{1}{2}\]× (10 + 30) × 103 × 15 × 10−6
= \[\frac{1}{2}\] × 40 × 15 × 10−3 = 0.30 J
Initial volume in path ADB, along line DB is the same as final volume. Thus, work done along this line is zero.
Along line AD, P = 10 kPa
W = WAD + WDB
= 10 × 103 (25 − 10) × 10−6 + 0
= 10 × 15 × 10−3 = 0.15 J
APPEARS IN
संबंधित प्रश्न
Explain why Two bodies at different temperatures T1 and T2, if brought in thermal contact, do not necessarily settle to the mean temperature (T1 + T2)/2.
Should the internal energy of a system necessarily increase if heat is added to it?
Should the internal energy of a system necessarily increase if its temperature is increased?
A closed bottle contains some liquid. the bottle is shaken vigorously for 5 minutes. It is found that the temperature of the liquid is increased. Is heat transferred to the liquid? Is work done on the liquid? Neglect expansion on heating.
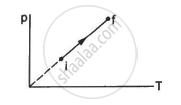
An ideal gas goes from the state i to the state f as shown in figure. The work done by the gas during the process ______________ .
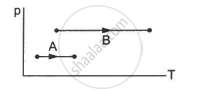
Consider two processes on a system as shown in figure.
The volumes in the initial states are the same in the two processes and the volumes in the final states are also the same. Let ∆W1 and ∆W2 be the work done by the system in the processes A and B respectively.
A gas is contained in a metallic cylinder fitted with a piston. The piston is suddenly moved in to compress the gas and is maintained at this position. As time passes the pressure of the gas in the cylinder ______________ .
In a process on a system, the initial pressure and volume are equal to the final pressure and volume.
(a) The initial temperature must be equal to the final temperature.
(b) The initial internal energy must be equal to the final internal energy.
(c) The net heat given to the system in the process must be zero.
(d) The net work done by the system in the process must be zero.
A 100 kg lock is started with a speed of 2.0 m s−1 on a long, rough belt kept fixed in a horizontal position. The coefficient of kinetic friction between the block and the belt is 0.20. (a) Calculate the change in the internal energy of the block-belt system as the block comes to a stop on the belt. (b) Consider the situation from a frame of reference moving at 2.0 m s−1 along the initial velocity of the block. As seen from this frame, the block is gently put on a moving belt and in due time the block starts moving with the belt at 2.0 m s−1. calculate the increase in the kinetic energy of the block as it stops slipping past the belt. (c) Find the work done in this frame by the external force holding the belt.
Which of the following is correct, when the energy is transferred to a system from its environment?
Which of the following system freely allows the exchange of energy and matter with its environment?
A system releases 100 kJ of heat while 80 kJ of work is done on the system. Calculate the change in internal energy.
The internal energy of a system is ______
8 m3 of a gas is heated at the pressure 105 N/m2 until its volume increases by 10%. Then, the external work done by the gas is ____________.
Two samples A and B, of a gas at the same initial temperature and pressure are compressed from volume V to V/2; A isothermally and B adiabatically. The final pressure of A will be ______.
An expansion process on a diatomic ideal gas (Cv = 5/2 R), has a linear path between the initial and final coordinates on a pV diagram. The coordinates of the initial state are: the pressure is 300 kPa, the volume is 0.08 m3 and the temperature is 390 K. The final pressure is 90 kPa and the final temperature s 320 K. The change in the internal energy of the gas, in SI units, is closest to:
If a gas is compressed adiabatically:
The internal energy of one mole of argon at 300 K is ______. (R = 8.314 J/mol.K)
A steam engine delivers 4.8 x 108 Jof work per minute and services 1.2 x 109 J of heat per minute from its boiler. What is the percentage efficiency of the engine?
A system releases 125 kJ of heat while 104 kJ work is done on the system. Calculate the change in internal energy.
