Advertisements
Advertisements
प्रश्न
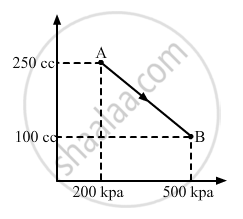
A gas is taken along the path AB as shown in figure. If 70 cal of heat is extracted from the gas in the process, calculate the change in the internal energy of the system.

Advertisements
उत्तर
Given:- 70 cal of heat is extracted from the system.
Here,
∆Q = -70 cal = -(70 × 4.2) J = -294 J
From the first law of thermodynamics, we get
∆W = P ∆ V
If P is the average pressure between points A and B and ∆V is the change in volume of the system while going from point A to B, then
∆W = `-1/2`× (200 + 500) × 103 × (150 × 10−6)
∆W = `-1/2`× 700 × 150 × 10−3
∆W = - 525 × 10−1 = - 52.5 J
Here, negative sign is taken because the final volume is less than the initial volume.
∆U = ?
∆Q = ∆U + ∆W
∆Q = - 294 J
Here, negative sign indicates that heat is extracted out from the system.
⇒ − 294 = ∆U - 52.5
⇒ ∆U = − 294 + 52.5 = - 241.5 J
APPEARS IN
संबंधित प्रश्न
Explain why Two bodies at different temperatures T1 and T2, if brought in thermal contact, do not necessarily settle to the mean temperature (T1 + T2)/2.
Explain why Air pressure in a car tyre increases during driving.
A closed bottle contains some liquid. the bottle is shaken vigorously for 5 minutes. It is found that the temperature of the liquid is increased. Is heat transferred to the liquid? Is work done on the liquid? Neglect expansion on heating.
Can work be done by a system without changing its volume?
Figure shows two processes A and B on a system. Let ∆Q1 and ∆Q2 be the heat given to the system in processes A and B respectively. Then ____________ .
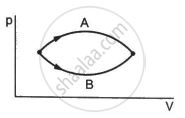
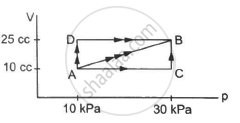
Figure shows three paths through which a gas can be taken from the state A to the state B. Calculate the work done by the gas in each of the three paths.
Which of the following is correct, when the energy is transferred to a system from its environment?
A system releases 100 kJ of heat while 80 kJ of work is done on the system. Calculate the change in internal energy.
Explain the different ways through which the internal energy of the system can be changed.
A thermodynamic system goes from states (i) P, V to 2P, V (ii) P, V to P, 2V. The work done in the two cases is ______.
In a thermodynamic system, working substance is ideal gas. Its internal energy is in the form of ______.
8 m3 of a gas is heated at the pressure 105 N/m2 until its volume increases by 10%. Then, the external work done by the gas is ____________.
Two samples A and B, of a gas at the same initial temperature and pressure are compressed from volume V to V/2; A isothermally and B adiabatically. The final pressure of A will be ______.
In insulated systems, the amount of external work done by the gas is proportional to:
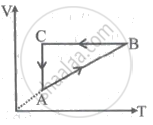
A cyclic process ABCA is shown in the V-T diagram. A process on the P-V diagram is ______.
The molar specific heat of He at constant volume is 12.47 J/mol.K. Two moles of He are heated at constant pressure. So the rise in temperature is 10 K. Find the increase in internal energy of the gas.
What is heat?
