Advertisements
Advertisements
प्रश्न
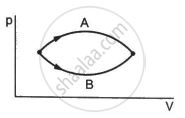
Figure shows two processes A and B on a system. Let ∆Q1 and ∆Q2 be the heat given to the system in processes A and B respectively. Then ____________ .
विकल्प
∆Q1 > ∆Q2
∆Q1 = ∆Q2
∆Q1 < ∆Q2
∆Q1 ≤ ∆Q2
Advertisements
उत्तर
∆Q1 > ∆Q2
Both the processes A and B have common initial and final points. So, change in internal energy, ∆U is same in both the cases. Internal energy is a state function that does not depend on the path followed.
In the P-V diagram, the area under the curve represents the work done on the system, ∆W. Since area under curve A > area under curve B, ∆W1> ∆W2.
Now,
∆Q1 = ∆U + ∆W1
∆Q2 = ∆U + ∆W2
But ∆W1 > ∆W2
⇒ ∆Q1 > ∆Q2
Here, ∆Q1 and ∆Q2 denote the heat given to the system in processes A and B, respectively.
APPEARS IN
संबंधित प्रश्न
Explain why Air pressure in a car tyre increases during driving.
Should the internal energy of a system necessarily increase if heat is added to it?
A force F is applied on a block of mass M. The block is displaced through a distance d in the direction of the force. What is the work done by the force on the block? Does the internal energy change because of this work?
Can work be done by a system without changing its volume?
An ideal gas goes from the state i to the state f as shown in figure. The work done by the gas during the process ______________ .
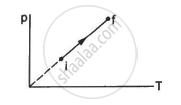
The pressure p and volume V of an ideal gas both increase in a process.
(a) Such a process is not possible.
(b) The work done by the system is positive.
(c) The temperature of the system must increase.
(d) Heat supplied to the gas is equal to the change in internal energy.
A substance is taken through the process abc as shown in figure. If the internal energy of the substance increases by 5000 J and a heat of 2625 cal is given to the system, calculate the value of J.

A gas is taken along the path AB as shown in figure. If 70 cal of heat is extracted from the gas in the process, calculate the change in the internal energy of the system.

A gas is initially at a pressure of 100 kPa and its volume is 2.0 m3. Its pressure is kept constant and the volume is changed from 2.0 m3 to 2.5 m3. Its Volume is now kept constant and the pressure is increased from 100 kPa to 200 kPa. The gas is brought back to its initial state, the pressure varying linearly with its volume. (a) Whether the heat is supplied to or extracted from the gas in the complete cycle? (b) How much heat was supplied or extracted?
Figure shows a cylindrical tube of volume V with adiabatic walls containing an ideal gas. The internal energy of this ideal gas is given by 1.5 nRT. The tube is divided into two equal parts by a fixed diathermic wall. Initially, the pressure and the temperature are p1, T1 on the left and p2, T2 on the right. The system is left for sufficient time so that the temperature becomes equal on the two sides. (a) How much work has been done by the gas on the left part? (b) Find the final pressures on the two sides. (c) Find the final equilibrium temperature. (d) How much heat has flown from the gas on the right to the gas on the left?

A system releases 100 kJ of heat while 80 kJ of work is done on the system. Calculate the change in internal energy.
The internal energy of a system is ______
8 m3 of a gas is heated at the pressure 105 N/m2 until its volume increases by 10%. Then, the external work done by the gas is ____________.
When 1 g of water at 0° C and 1 x 105 N/m2 pressure is converted into ice of volume 1.082 cm3, the external work done will be ____________.
A person of mass 60 kg wants to lose 5kg by going up and down a 10 m high stairs. Assume he burns twice as much fat while going up than coming down. If 1 kg of fat is burnt on expending 7000 kilo calories, how many times must he go up and down to reduce his weight by 5 kg?
A steam engine delivers 4.8 x 108 Jof work per minute and services 1.2 x 109 J of heat per minute from its boiler. What is the percentage efficiency of the engine?
