Advertisements
Advertisements
Write the IUPAC name of the following compound and classify it as primary, secondary and tertiary amine.
(CH3CH2)2NCH3
Concept: Nomenclature of Amines
Account for the following:
Methylamine in water reacts with ferric chloride to precipitate hydrated ferric oxide.
Concept: Nomenclature of Amines
Arrange the following:
In increasing order of basic strength:
C6H5NH2, C6H5NHCH3, C6H5CH2NH2
Concept: Chemical Reactions of Amines - Basic Character of Amines
Write a short note on the following:
Hoffmann’s bromamide reaction
Concept: Preparation of Amines
Write the structure of 2,4-dinitrochlorobenzene
Concept: Nomenclature of Amines
Give reasons for the following:
CH3NH2 is more basis than C6H5NH2.
Concept: Classification of Amines
Write the IUPAC name of the given compound :
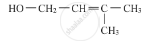
Concept: Nomenclature of Amines
Illustrate the following reactions giving suitable example in each case
Ammonolysis
Concept: Physical Properties of Amines
Illustrate the following reactions giving suitable example in each case
Coupling reaction
Concept: Chemical Reaction of Diazonium Salts - Reactions Involving Retention of Diazo Group
Illustrate the following reactions giving suitable example in each case
Acetylation of amines
Concept: Chemical Reactions of Amines - Electrophilic Substitution
Give the structures of A, B and C in the following reactions :
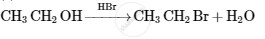
Concept: Preparation of Amines
Give the structures of A, B and C in the following reactions :

Concept: Preparation of Amines
Give the structures of A, B and C in the following reactions :

Concept: Preparation of Amines
Arrange the following in increasing order of basic strength :
C6H5NH2, C6H5NHCH3, C6H5N(CH3)2
Concept: Physical Properties of Amines
Draw the structures of the following compounds :
N2O5
Concept: Chemical Reactions of Amines - Reaction with Nitrous Acid
Account for the following:
Gabriel phthalimide synthesis is not preferred for preparing aromatic primary amines.
Concept: Preparation of Amines
Account for the following:
On reaction with benzene sulphonyl chloride, primary amine yields product soluble in alkali whereas secondary amine yields product insoluble in alkali.
Concept: Chemical Reactions of Amines - Reaction with Arylsulphonyl Chloride
Write structures of compounds A and B in each of the following reactions: 
Concept: Structure of Amines
Write structures of compounds A and B in each of the following reactions:
Concept: Preparation of Amines
Write chemical equation in support of your answer.
Out of 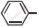 Cl and O2N
Cl and O2N  Cl. which one is more reactive towards nucleophilic substitution reaction and why?
Cl. which one is more reactive towards nucleophilic substitution reaction and why?
Concept: Chemical Reactions of Amines - Reaction with Arylsulphonyl Chloride
