Advertisements
Advertisements
Question
Complete the following equations:
KMnO4 
Advertisements
Solution
2 KMnO4 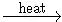 K2MnO4 + MnO2 + O2
K2MnO4 + MnO2 + O2
APPEARS IN
RELATED QUESTIONS
Draw structure of Chlorine pentafluoride
Fluorine is a stronger oxidising agent than chlorine. Why?
Account for the following :
Acidic character increases from HF to HI.
Arrange the following in the order of property indicated for the given set:
HF, HCl, HBr, HI - increasing acid strength.
Complete chemical reactions are:Cl2 + H2O →
Tincture of iodine is ____________.
Which one has the lowest boiling point?
The set with the correct order of acidity is:
Which of the following statements are true?
(i) Only type of interactions between particles of noble gases are due to weak dispersion forces.
(ii) Ionisation enthalpy of molecular oxygen is very close to that of xenon.
(iii) Hydrolysis of XeF6 is a redox reaction.
(iv) Xenon fluorides are not reactive.
Give a reason for the following:
Mn+2 compounds are more stable than Fe+2 compounds.
