Advertisements
Advertisements
प्रश्न
Complete the following equations:
KMnO4 
Advertisements
उत्तर
2 KMnO4 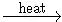 K2MnO4 + MnO2 + O2
K2MnO4 + MnO2 + O2
APPEARS IN
संबंधित प्रश्न
Iodine exists as
- polar molecular solid
- ionic solid
- nonpolar molecular solid
- hydrogen bonded molecular solid
Compare the oxidizing action of F2 and Cl2 by considering parameters such as bond dissociation enthalpy, electron gain enthalpy and hydration enthalpy.
F2 has lower bond dissociation enthalpy than Cl2. Why?
Which halogen compound in each of the following pairs will react faster in SN2 reaction
(CH3)3 C – Cl or CH3 – Cl
Write two uses of ClO2.
With what neutral molecule is ClO− isoelectronic? Is that molecule a Lewis base?
Which is the strongest acid in the following:
Which of the following pairs of ions are isoelectronic and isostructural?
In solid state \[\ce{PCl5}\] is a ______.
Which of the following statements are true?
(i) Only type of interactions between particles of noble gases are due to weak dispersion forces.
(ii) Ionisation enthalpy of molecular oxygen is very close to that of xenon.
(iii) Hydrolysis of XeF6 is a redox reaction.
(iv) Xenon fluorides are not reactive.
