Definitions [31]
Long answer question.
What is metabolism?
Metabolism is the sum of the chemical reactions that take place within each cell of a living organism and provide energy for vital processes and for synthesizing new organic material.
Long answer question.
What are the nucleic acids?
Nucleic acids are macromolecules composed of many small units or monomers called nucleotides.
Long answer question.
What are biomolecules?
Biomolecules are essential substances produced by our body which are necessary for life.
Define carbohydrates.
Carbohydrates are optically active polyhydroxy aldehydes or polyhydroxy ketones or compounds that can be hydrolysed to polyhydroxy aldehydes or polyhydroxy ketones.
Carbohydrates that are crystalline solids, sweet in taste and soluble in water are called sugars.
Carbohydrates that are amorphous solids, tasteless and insoluble in water are catled non-sugars.
The sugars that reduce the Tollen's reagent and Fehling's solution are called reducing sugars.
Monosaccharides that contains one aldehydic group is called aldose.
A ketose with six carbon atoms is called a ketohexose.
An aldose monosaccharide that has six carbon atoms (e.g., Glucose) is called an aldohexose.
A monosaccharide that contains one ketonic carbonyl group is called a ketose.
Define the term Protein.
Chemically proteins are polyamides which are high molecular weight polymers of the monomer units, i.e., α-amino acids. OR It can also be defined as proteins are the biopolymers of a large number of α-amino acids and they are naturally occurring polymeric nitrogenous organic compounds containing 16% nitrogen and peptide linkages (-CO-NH-)
Bifunctional organic compounds containing a carboxylic and an amino group either at the same carbon atom or at nearby carbon atoms are called amino acids.
Chemically, proteins are polyamides, which are high molecular weight polymers of the monomer units called \[\alpha\]-amino acids.
Define peptide bond.
The bond that connects α-amino acids to each other is called a peptide bond.
Define α-amino acids.
α-Amino acids are carboxylic acids having an amino (–NH2) group bonded to the α-carbon, that is, the carbon next to the carboxyl (–COOH) group.
Define enzymes.
Enzymes are biological catalysts that speed up chemical reactions in living cells without being consumed in the process.
Amino acids which contain more number of carboxyl groups than amino groups are called acidic amino acids.
Amino acids which contain equal number of amino groups and carboxyl groups are called neutral amino acids.
Amino acids which contain more number of amino groups than carboxyl groups are called basic amino acids.
Amino acids which are synthesised by the body itself are called non-essential amino acids.
Define the following term as related to proteins:
Primary structure
Proteins may have one or more polypeptide chains. Each polypeptide in a protein has amino acids linked with each other in a specific sequence and it is this sequence of amino acids that is said to be the primary structure of that protein. Any change in this primary structure, i.e., the sequence of amino acids, creates a different protein.
Define the following as related to proteins:
Peptide linkage
Chemically, peptide linkage is an amide formed between the –COOH group and –NH2 group. The reaction between two molecules of similar or different amino acids proceeds through the combination of the amino group of one molecule with the carboxyl group of the other. This results in the elimination of a water molecule and the formation of a peptide bond –CO–NH–. The product of the reaction is called a dipeptide because it is made up of two amino acids.
For example, when the carboxyl group of glycine combines with the amino group of alanine, we get a dipeptide, glycylalanine.
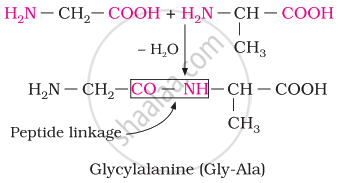
Define the following term as related to proteins:
Denaturation
Denaturation is the process in which the secondary and tertiary structure of a protein is disrupted due to heat, a change in pH, or chemicals, while the primary structure remains unchanged. In denaturation, peptide bonds are not broken; only the weak bonds (like hydrogen bonds) are disturbed.
Nucleic acids are large biological macromolecules that store and transmit genetic information in living organisms.
DNA is a double-stranded nucleic acid that stores and transmits hereditary information and can replicate itself.
RNA is a single-stranded nucleic acid that helps in protein synthesis and information transfer.
A nucleotide is the basic structural unit of nucleic acids, composed of a nitrogenous base, a pentose sugar, and a phosphate group.
A nucleoside consists of a nitrogenous base linked to a pentose sugar without a phosphate group.
Define the following term.
nucleoside
The unit formed by joining the anomeric carbon of the furanose (sugar) with a nitrogen of a base is called nucleoside.
A nitrogenous base is an organic molecule (purine or pyrimidine) that carries genetic information in nucleic acids.
Key Points
- Carbohydrates are organic biomolecules made of C, H and O, usually fitting the general formula Cx(H₂O)y and existing as aldoses or ketoses.
- They are classified into monosaccharides, disaccharides and polysaccharides; monosaccharides cannot be hydrolysed further, disaccharides are formed by two monosaccharides via glycosidic bonds, and polysaccharides are long polymers.
- Some sugars like digitoxose (C₆H₁₂O₄) and rhamnose (C₆H₁₂O₅) do not obey the typical Cx(H₂O)y formula.
- All monosaccharides are reducing sugars because they possess a free aldehyde or ketone group.
- Cellulose is a linear polymer of β‑D‑glucose, unlike starch and glycogen, which are polymers of α‑glucose and show branching.
- Biologically, carbohydrates supply energy for metabolism; glucose is the main substrate for ATP synthesis, and lactose provides energy to infants.
- Polysaccharides such as starch and glycogen act as storage products and also contribute to structural components of cell membranes and cell walls.
- Glucose is a monosaccharide, an aldohexose, and a reducing sugar, commonly found in fruits and also known as dextrose.
- It can be prepared by hydrolysis of sucrose (using dilute acid) or hydrolysis of starch under heat and pressure.
- Glucose confirms a straight-chain structure of six carbon atoms when reduced to n-hexane.
- Presence of functional groups is shown by reactions: –CHO (aldehyde), five –OH groups, and formation of derivatives like oxime and cyanohydrin.
- Oxidation reactions indicate the formation of gluconic acid (mild oxidation) and saccharic acid (strong oxidation), confirming functional groups in glucose.
| Product | Inference |
|---|---|
| n-Hexane (hot HI) | 6 C in a straight chain |
| Glucoxime (NH₂OH) / Cyanohydrin (HCN) | Carbonyl group present |
| Gluconic acid (Br₂ water) | —CHO group present |
| Glucose pentaacetate (acetic anhydride) | Five —OH groups present |
| Saccharic acid (dil. HNO₃) | One primary —OH group present |
- Glucose is an aldohexose with molecular formula \[C_{6}H_{12}O_{6},\mathrm{M.P.146^{\circ}C.}\]
- 'D' in D-(+)-Glucose = configuration; (+) = dextrorotatory nature; 'D'/'L' have no relation to optical activity.
- Glucose has five —OH groups (confirmed by glucose pentaacetate) and one aldehydic carbonyl group (confirmed by oxime & cyanohydrin formation).
- Glucose is soluble in water, sparingly soluble in alcohol, and insoluble in ether.
- The additional chiral centre in glucose ring structures is formed due to ring closure.
| Open Chain Structure | Ring Structure | |
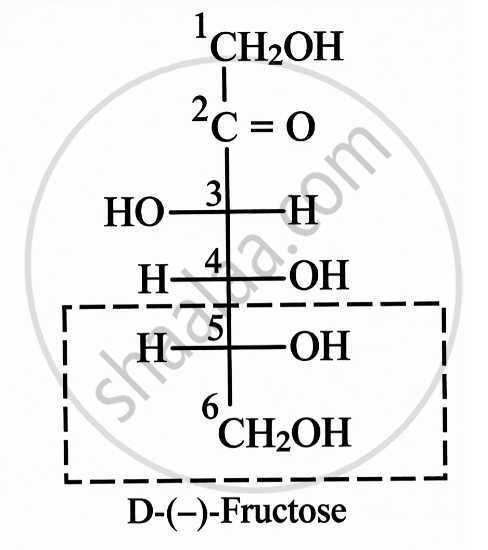 |
\[\alpha\]-D-(-)-Fructofuranose | \[\beta\]-D-(-)-Fructofuranose |
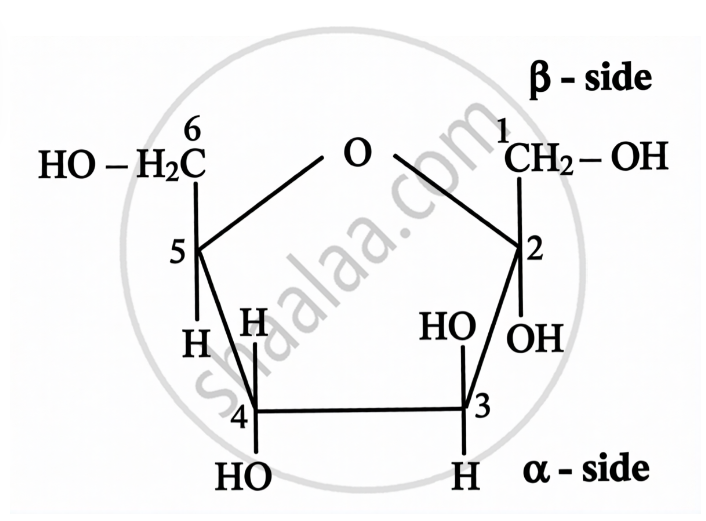 |
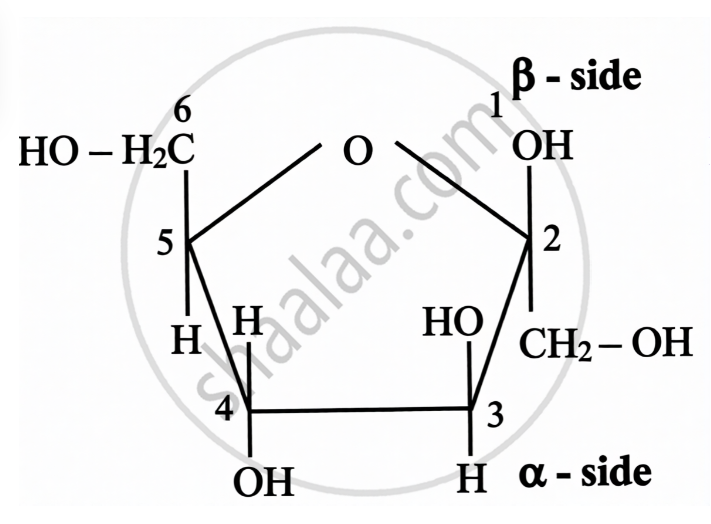 |
|
- Sucrose consists of one unit each of α-D-glucopyranose and β-D-fructofuranose.
- It contains an α, β-1,2-glycosidic linkage.
- Maltose is composed of two α-D-glucopyranose units joined by an α-1,4-glycosidic bond.
- Lactose consists of β-D-galactopyranose and β-D-glucopyranose units.
- It has a β-1,4-glycosidic linkage.
- Starch — A polymer of α-D-glucopyranose. It has two components: amylose (α-1,4-glycosidic linkage) and amylopectin (both α-1,4 and α-1,6-glycosidic linkages).
- Cellulose — A polymer of β-glucopyranose units linked by β-1,4-glycosidic bonds.
- Glycogen — A polymer of glucose units.
- Linkage Comparison —
Polysaccharide Monomer Linkage Amylose (Starch) α-D-glucopyranose α-1,4 Amylopectin (Starch) α-D-glucopyranose α-1,4 and α-1,6 Cellulose β-glucopyranose β-1,4 Glycogen Glucose — - Key Distinction — Starch and Cellulose are both glucose polymers but differ in linkage type: Starch has α-glycosidic bonds (digestible by humans), while Cellulose has β-glycosidic bonds (not digestible by humans).
- Origin of the name: The word "protein" is derived from the Greek word 'proteios', meaning "primary" or "of prime importance", reflecting proteins' essential role in living organisms.
- Chemical nature: Proteins are polyamides — high molecular weight polymers made up of monomer units called α-amino acids (general formula: RCH(NH₂)COOH).
- Nutritional sources: Key dietary sources of protein include milk, pulses, nuts, fish, and meat.
- Fibrous proteins have thread-like structures where polypeptide chains run parallel, held by hydrogen and disulphide bonds; they are insoluble in water — e.g., Keratin (hair, skin, nails) and Myosin (muscles).
- Globular proteins have a spherical shape where polypeptide chains coil around; they are soluble in water — e.g., Insulin, Egg albumin, and Legumelin.
| Sr. No. | Name | R Group | 3-Letter | 1-Letter |
|---|---|---|---|---|
| 1 | Glycine | H– | Gly | G |
| 2 | Alanine | CH₃– | Ala | A |
| 3 | Valine* | Me₂CH– | Val | V |
| 4 | Leucine* | Me₂CH–CH₂– | Leu | L |
| 5 | Isoleucine* | CH₃–CH₂–CH(Me)– | Ile | I |
| 6 | Asparagine | H₂N–CO–CH₂– | Asn | N |
| 7 | Glutamine | H₂N–CO–CH₂–CH₂– | Gln | Q |
| 8 | Serine | HO–CH₂– | Ser | S |
| 9 | Threonine* | CH₃–CHOH– | Thr | T |
| 10 | Cysteine | HS–CH₂– | Cys | C |
| 11 | Methionine* | Me–S–CH₂–CH₂– | Met | M |
| 12 | Phenylalanine* | Ph–CH₂– | Phe | F |
| 13 | Tyrosine | p–HO–C₆H₄–CH₂– | Tyr | Y |
| 14 | Tryptophan* | Indole–CH₂– | Trp | W |
| 15 | Proline | Entire ring structure | Pro | P |
| 16 | Aspartic acid (Acidic) | HOOC–CH₂– | Asp | D |
| 17 | Glutamic acid (Acidic) | HOOC–CH₂–CH₂– | Glu | E |
| 18 | Lysine* (Basic) | H₂N–(CH₂)₄– | Lys | K |
| 19 | Arginine* (Basic) | HN=C(NH₂)–NH–(CH₂)₃– | Arg | R |
| 20 | Histidine* (Basic) | Imidazole–CH₂– | His | H |
- Proteins are made up of α-amino acids linked together in a chain.
- A peptide bond (–CO–NH–) is formed between the carboxyl group of one amino acid and the amino group of another with the removal of water (condensation).
- Two amino acids form a dipeptide, three form a tripeptide, and many form polypeptides.
- Proteins are long polypeptide chains containing more than 100 amino acid residues.
- The ends of a protein chain are different:
The N-terminal has a free amino group, and the C-terminal has a free carboxyl group. - A peptide bond is similar to a secondary amide linkage in organic chemistry.
- The primary structure of proteins shows the sequence of amino acids in a polypeptide chain.
- Secondary structure is formed by hydrogen bonding and mainly includes α-helix and β-pleated sheet.
- In an α-helix, the chain coils into a right-handed spiral stabilised by hydrogen bonds.
- In a β-pleated sheet, chains are stretched and arranged side by side, held by intermolecular hydrogen bonds.
- Tertiary structure is the overall 3D folding of the chain due to interactions like hydrogen bonds, disulfide bonds, and van der Waals forces, while quaternary structure is the arrangement of multiple polypeptide chains.
1. Enzymes are biological (protein) catalysts that increase the rate of biochemical reactions without being consumed.
2. Most enzymes are globular proteins and show high specificity due to their unique 3D structure and active site.
3. Enzymes bind with substrate to form enzyme–substrate complex, then release product and remain unchanged.
4. Enzyme activity is affected by temperature and pH, and reactions are usually reversible.
5. Types of enzymes:
- Simple enzymes (only protein)
- Conjugated enzymes (protein + cofactor)
6. Examples of enzymes and their functions:
- Amylase: Starch → glucose
- Maltase: Maltose → glucose
- Lactase: Lactose → glucose + galactose
- Invertase: Sucrose → glucose + fructose
- Pepsin: Proteins → amino acids
- Nucleic acids are biomolecules that carry genetic information in cells (DNA and RNA).
- On hydrolysis, they give three components: pentose sugar, phosphoric acid, and nitrogenous bases.
- DNA contains β-D-2-deoxyribose, while RNA contains β-D-ribose.
- Nitrogenous bases include: Adenine, Guanine, Cytosine, Thymine (in DNA), and Uracil (in RNA).
- DNA is a double-stranded helix with antiparallel strands and contains A, T, G, C.
- RNA is single-stranded, contains A, G, C, U, and helps in protein synthesis.
- Nitrogenous bases in nucleic acids are of two types: purines and pyrimidines.
- Purine bases have a double-ring structure and include Adenine (A) and Guanine (G).
- Pyrimidine bases have a single-ring structure and include Cytosine (C), Thymine (T), and Uracil (U).
- Thymine is present in DNA, while Uracil is present in RNA instead of thymine.
Concepts [17]
- Biomolecules in the Cell
- Biomolecules in the Cell > Carbohydrates
- Monosaccahrides
- Preparation of Glucose
- Structures of Glucose
- Fructose
- Disaccharides
- Polysaccharides
- Biomolecules in the Cell > Proteins
- Classification of Amino Acids
- Peptide
- Structure of Proteins
- Denaturation of Proteins
- Biomolecules in the Cell > Enzymes
- Mechanism of Enzymatic Action
- Biomolecules in the Cell > Nucleic Acids
- Structure of Nucleic Acids
