Definitions [27]
A solution in which no solute can be dissolved further at a given temperature is called a saturated solution.
A solution in which more solute can be dissolved without raising the temperature is called an unsaturated solution.
Define isotonic solutions.
Two or more solutions exerting the same osmotic pressure are called isotonic solutions.
When two solutions are separated by a semipermeable membrane and no osmosis occurs, i.e., there is no net flow of water on either side through the membrane, the solutions are said to be isotonic solutions. If the membrane is perfectly semipermeable, the two solutions possess the same osmotic pressure and are also referred to as iso-osmotic solutions.
Define the term:
Ideal solution
It is characterised as a solution that adheres to Raoult's Law, with no interactions between the molecules and no volume or heat change during mixing.
For an ideal solution, Enthalpy of mixing of the pure components to form the solution is Δmix H = 0 and the volume of mixing is Δmix V = 0.
A solution which contains more solute than would be necessary to saturate it at a given temperature is called a supersaturated solution.
Define mass percentage.
The mass percentage of a component of a solution is defined as the mass of the solute in grams present in 100 g of the solution.
Define molarity (M).
Molarity (M) is defined as the number of moles of solute dissolved in one litre (or one cubic decimetre) of solution.
Mole fraction of a constituent is the fraction obtained by dividing the number of moles of that constituent by the total number of moles of all the constituents present in the solution.
\[x_{1}=\frac{n_{1}}{n_{1}+n_{2}+n_{3}+...+n_{i}}\]
Define mole fraction.
The mole fraction of a particular component in a solution is the ratio of the number of moles of that component to the total number of moles of all the components present in the solution.
Define molality.
Molality (m) is defined as the number of moles of the solute dissolved in one kilogram (kg) of the solvent. The units of molality are moles per kilogram, i.e., mole kg−1. The molality is preferred over molarity if the volume of the solution is either expanding or contracting with temperature.
Molality (m) = `"Number of mole of solute"/"Mass of solvent (in kg)"`
Define Normality.
Normality (N) of a solution is defined as the number of gram equivalents of the solute present in one liter of the solution. Normality is used in acid-based redox titrations.
Normality (N) = `"Number of gram equivalents of solute"/"Volume of solution in litre"`
Define Freezing point.
The temperature at which the liquid and solid forms of a substance can exist together in equilibrium is called the freezing point of that substance.
Define Cryoscopic constant.
Cryoscopic constant or the Molal depression constant is defined as the depression in freezing point when one mole of non-volatile solute is dissolved in one kilogram of solvent. Its unit is K Kg mol−1.
Osmotic pressure is the minimum pressure which needs to be applied to a solution to prevent the inward flow of its pure solvent across a semipermeable membrane.
\[\pi=\frac{n_2RT}{V}=\mathrm{CRT}\]
\[\pi=\frac{w_2RT}{\mathrm{M}_2V}\]
Define Osmosis.
The net spontaneous flow of solvent molecules into the solution or from more dilute solution to more concentrated solution through a semipermeable membrane is called osmosis.
Define the following term:
Hypotonic solution
The solution having lower osmotic pressure as compared to some other solution is referred to as a hypotonic solution.
Define osmotic pressure.
Osmotic pressure may be defined as the external pressure which should be applied to the solution in order to stop the phenomenon of osmosis, i.e., to stop the flow of solvent into the solution when the two are separated by a semipermeable membrane.
Define Semipermeable membrane
Semipermeable membrane: It is a membrane which allows the solvent molecules, but not the solute molecules, to pass through it.
Semipermeable membrane is a film such as cellophane which has pores large enough to allow the solvent molecules to pass through them.
Define the following term:
isotonic solution
Two or more solutions exerting the same osmotic pressure are called an isotonic solution.
Define reverse osmosis.
The process of moving a solvent from a solution to a pure solvent through a semipermeable membrane while applying excessive pressure on the solution side is known as reverse osmosis.
It is a thin film, such as cellophane, which has pores large enough to allow the solvent molecules to pass through them.
or
When a solution and pure solvent or two solutions of different concentrations are separated by a semipermeable membrane, the solvent molecules pass through the membrane this is called osmosis.
It is the net spontaneous flow of solvent molecules into the solution or from a more dilute solution to a more concentrated solution through a semipermeable membrane.
If a pressure larger than the osmotic pressure is applied to the solution side, then pure solvent from the solution passes into the pure solvent side through the semipermeable membrane. This phenomenon is called reverse osmosis.
or
Osmosis is a flow of solvent through a semipermeable membrane into the solution. The direction of osmosis can be reversed by applying a pressure larger than the osmotic pressure. This is called reverse osmosis.
Define van’t Hoff factor.
The ratio of the observed (experimental) value of a colligative property to the normal (calculated) value of the same property is termed as van’t Hoff factor, i.
Define the term abnormal molar mass.
When the molar mass calculated using colligative properties differs from the theoretical molar mass, it is called an abnormal molar mass.
Define the following term:
Colligative properties
Colligative Properties: Colligative properties are the properties of the solutions which depend upon the number of solute particles present in the solution, irrespective of their nature, relative to the total number of particles present in the solution.
Examples: Relative lowering of vapour pressure of the solvent, depression of freezing point of the solvent, elevation of boiling point of the solvent, osmotic pressure of the solution
Define the following term:
Molal elevation constant (Kb)
Molal elevation constant (Kb) is defined as the elevation in boiling point of a solution when one mole of a non-volatile solute is dissolved in one kilogram of a volatile solvent.
Formulae [3]
\[\mathrm{Molarity}=\frac{\text{Number of moles of solute}}{\text{Volume of solution in litres}}\]
\[=\frac{w}{\mathrm{Molar~Mass}}\times\frac{1000}{V}\]
\[\mathrm{Normality}=\frac{\text{Number of gram equivalents of solute}}{\text{Volume of solution of litres}}\]
Normality of a solution = Molarity × nf
\[\mathrm{Molality~}=\frac{\text{Number of moles of solute}}{\text{Mass of solvent in kg}}=\frac{W_{\mathrm{B}}}{M_{\mathrm{B}}}\times\frac{1000}{W_{\mathrm{A}}}\]
where A = solvent, B = solute
Theorems and Laws [1]
State Henry’s law.
Henry’s Law states that at a constant temperature, the solubility of a gas in a liquid is directly proportional to the partial pressure of that gas above the liquid.
- Henry was the first to give a quantitative relationship between the pressure and solubility of a gas in a solvent, which is known as Henry’s law. The law states that at a constant temperature, the solubility of a gas in a liquid is directly proportional to the partial pressure of the gas present above the surface of the liquid or solution.
- Dalton, a contemporary of Henry, also concluded independently that the solubility of a gas in a liquid solution is a function of the partial pressure of the gas. If we use the mole fraction of a gas in the solution as a measure of its solubility, then it can be said that the mole fraction of gas in the solution is proportional to the partial pressure of the gas over the solution.
- The most commonly used form of Henry’s law states that “the partial pressure of the gas in the vapour phase (p) is proportional to the mole fraction of the gas (x) in the solution” and is expressed as:
p ∝ x
p = KH . x - Here, KH is Henry’s law constant. When a mixture of more than one gas is brought into contact with a solvent, each gaseous component dissolves in proportion to its partial pressure. That is why Henry’s law is applied to every gas, independent of the presence of other gases.
Key Points
Based on the physical states of solute and solvent, there are 9 types of solutions:
| S.No. | Solute | Solvent | Example |
|---|---|---|---|
| 1 | Solid | Liquid | Sea water, sugar in water, benzoic acid in benzene |
| 2 | Solid | Solid | Alloys — brass, bronze |
| 3 | Solid | Gas | Iodine in air |
| 4 | Liquid | Liquid | Ethanol in water, gasoline |
| 5 | Liquid | Solid | Amalgam (mercury in silver) |
| 6 | Liquid | Gas | Chloroform in nitrogen |
| 7 | Gas | Liquid | Carbonated water (CO₂ in water), O₂ in water |
| 8 | Gas | Solid | H₂ in palladium |
| 9 | Gas | Gas | Air (O₂, N₂, Ar mixture) |
The relative lowering of vapour pressure of a solution containing a non-volatile solute is equal to the mole fraction of the solute in the solution.
\[\frac{p^\circ-p_{\mathrm{solution}}}{p^\circ}=\frac{n_2}{n_1+n_2}\]
Freezing point of solution is smaller than that of pure solvent and is given by
ΔTf = Kf × m
Kf = Metal depression constant or cryoscopic constant
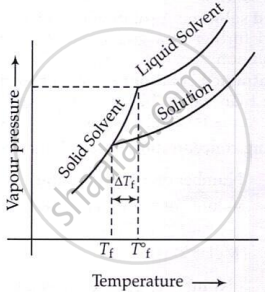
where, \[\Delta T_{\mathbf{f}}=T_{\mathbf{f}}^{\circ}-T_{\mathbf{f}}\]
\[\Delta T_\mathrm{f}=\frac{\mathrm{K}_f\times W_2\times1000}{M_2\times W_1}\]
Boiling point of solution is greater than that of pure solvent and is given by
ΔTb = Kb × m
- Kb = Metal elevation constant or Ebullioscopic constant
- M = molality
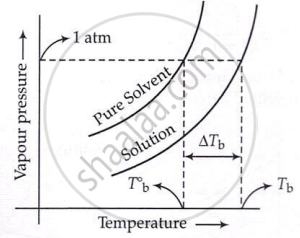
where,
\[\Delta T_{\mathbf{b}}=T_{\mathbf{b}}-T_{\mathbf{b}}^{\circ}\]
\[\Delta T_{\mathrm{b}}=\frac{K_{\mathrm{b}}\times W_{2}\times1000}{M_{2}\times W_{1}}\]
Important Questions [40]
- Define isotonic solutions.
- Define Boiling Point
- Derive Van’T Hoff General Solution Equation
- Define molality.
- Why is Molality of a Solution Independent of Temperature?
- When Koh Solution is Added to Potassium Dichromate Solution the Colour of Solution Changes to Yellow, Because
- 22.22 gram of urea was dissolved in 300 grams of water. Calculate the number of moles of urea and molality of the urea solution. (Given: a Molar mass of urea = 60 gram mol-1)
- How does solubility of a gas in water varies with the temperature?
- Which Mixture is Used for Respiration by Deep Sea Divers?
- State Henry’s law.
- What is the effect of temperature on solubility of a gas in a liquid?
- Explain, Why Do Aquatic Animals Prefer to Stay at Lower Level of Water During Summer?
- Derive the Relation ∆H − ∆U = ∆Nrt.
- The Boiling Point of Water at High Altitude is Low. Because
- Derive the relationship between relative lowering of vapour pressure and molar mass of nonvolatile solute.
- The Temperature at Which Vapour Pressure of a Liquid Becomes Equal to the Atmospheric Pressure is
- Define Freezing point.
- Which of the Following Solutions Shows Maximum Depression in Freezing Point?
- Define Cryoscopic constant.
- What is the Freezing Point of a Liquid? the Freezing Point of Pure Benzene is 278.4 K. Calculate the Freezing Point of the Solution When 2.0 G of a Solute Having Molecular Weight 100 G Mol-1
- Write the Formula to Determine the Molar Mass of a Solute Using Freezing Point Depresssion Method.
- 1.0 x10-3Kg of urea when dissolved in 0.0985 Kg of a solvent, decreases freezing point of the solvent by 0.211 k.
- Define osmotic pressure.
- Which of the Following is Not a Colligative Property?
- Which of the following 0.1 M aqueous solutions will exert the highest osmotic pressure?
- Choose the most correct option. In calculating osmotic pressure the concentration of solute is expressed in _______.
- Answer the following in one or two sentences. What is osmotic pressure?
- Define Osmosis.
- Derive an expression to calculate molar mass of non-volatile solute by osmotic pressure measurement.
- How will you determine molar mass of solute from osmotic pressure?
- Write the condition of reverse osmosis.
- Define Semipermeable Membrane. OR Explain the term semipermeable membrane.
- Define the Following Term: Isotonic Solution
- Define the following term: Hypotonic solution
- How Van’T Hoff Factor is Related to the Degree of Dissociation?
- The substance ‘X’, when dissolved in solvent water gave molar mass corresponding to the molecular formula ‘X3’. The van’t Hoff factor (i) is
- Derive van’t Hoff general solution equation.
- Define van’t Hoff factor.
- Derive the Relation Between the Elevation of Boiling Point and Molar Mass of Solute.
- Define the following term: Colligative properties
Concepts [10]
- Types of Solutions
- Expressing Concentration of Solutions
- Solubility of a Gas in a Liquid
- Solubility of a Solid in a Liquid
- Colligative Properties and Determination of Molar Mass
- Relative Lowering of Vapour Pressure
- Depression of Freezing Point
- Osmosis and Osmotic Pressure
- Abnormal Molar Masses
- Elevation of Boiling Point
