(English Medium)
Academic Year: 2018-2019
Date: March 2019
Advertisements
1) Attempt all questions from section 1
2) Attempt any four questions from section 2
Choose the correct answer from the options given below :
An electrolyte which completely dissociates into ions are :
Alcohol
Carbonic acid
Sucrose
Sodium hydroxide
Chapter: [3.2] Salts and their Preparations
The most electronegative element from the following elements is:
Magnesium
Chlorine
Aluminium
Sulphur
Chapter:
The reason for using Aluminium in the alloy duralumin is ______.
Aluminium is brittle
Aluminium gives strength
Aluminium brings lightness
Aluminium lowers melting point
Chapter:
The drying agent used to dry HCl gas is ______.
Conc. H2SO4
ZnO
Al2O3
CaO
Chapter:
A hydrocarbon which is a greenhouse gas is ______.
Acetylene
Ethylene
Ethane
Methane
Chapter:
Conversion of ethanol to ethene by the action of concentrated sulphuric acid is an example of _________.
Dehydration
Dehydrogenation
Dehydrohalogenation
Hydrolysis
Chapter:
When sodium chloride is heated with concentrated sulphuric acid below 200°C, one of the products formed is ______.
sodium hydrogen sulphate
sodium sulphate
chlorine
Chapter: [8] Study of Compounds A. Hydrogen Chloride
Ammonia reacts with excess chlorine to form ______.
nitrogen
nitrogen trichloride
ammonium chloride
Chapter: [9] Study of Compounds B. Ammonia
Fill in the blanks with the choices given in brackets :
Substitution reactions are characteristic reactions of ____________ (alkynes / alkenes / alkanes)
Chapter:
In Period 3, the most metallic element is ______.
sodium
magnesium
aluminium
Chapter:
Write a balanced chemical equation for each of the following reactions :
(i) Reduction of copper (II) oxide by hydrogen
Chapter: [7.2] Stages Involved in the Extraction of Metals
Write a balanced chemical equation for each of the following reactions :
Action of dilute sulphuric acid on sodium hydroxide.
Chapter: [11] Study of Compounds D. Sulphuric Acid
Write a balanced chemical equation for each of the following reactions :
Action of dilute sulphuric acid on zinc sulphide.
Chapter: [11] Study of Compounds D. Sulphuric Acid
Write a balanced chemical equation for each of the following reactions :
Ammonium hydroxide is added to ferrous sulphate solution.
Chapter: [9] Study of Compounds B. Ammonia
Write a balanced chemical equation for each of the following reactions :
Chlorine gas is treated with ethane.
Chapter:
State one observation for the following:
Concentrated nitric acid is reacted with sulphur.
Chapter: [10] Study of Compounds C. Nitric Acid
Ammonia gas is passed over heated copper (I) oxide.
Chapter: [9] Study of Compounds B. Ammonia
State one observation for each of the following :
Copper sulphate solution is electrolysed using copper electrodes.
Chapter:
State one observation for each of the following :
A small piece of zinc is added to dilute hydrochloric acid
Chapter: [3.1] Acids and Bases
State one observation for each of the following :
Lead nitric is heated strongly in a test tube.
Chapter: [3.2] Salts and their Preparations
Calculate the number of moles in 12g of oxygen gas.
Chapter: [5.2] Relative Atomic Mass, Relative Molecular Mass and Mole Concept
Calculate :
The weight of 1022 atoms of carbon [C = 12, Avogadro’s No. = `6xx10^23 ]`
Chapter:
Give the IUPAC name of the following organic compounds :
Chapter:
What is the special feature of the structure of ethyne?
Chapter:
Name the saturated hydrocarbon containing two carbon atoms.
Chapter:
Give the structural formula of Acetic acid.
Chapter: [12.6] Carboxylic Acids
Advertisements
Give the appropriate term defined by the statements given below :
The formula that represents the simplest ratio of the various elements present in one molecule of the compound.
Chapter: [5.3] Percentage Composition, Empirical and Molecular Formulae
The tendency of an atom to attract electrons itself when combined in a covalent compound.
Chapter:
Give the appropriate term defined by the statements given below :
The substance that releases hydronium ion as the only positive ion when dissolved in water.
Chapter: [3.1] Acids and Bases
The tendency of an atom to attract electrons itself when combined in a covalent compound.
Chapter:
The covalent bond in which the electrons are shared equally between the combining atoms.
Chapter: [2] Chemical Bonding
The covalent bond in which the electrons are shared equally between the combining atoms.
Chapter: [2] Chemical Bonding
K, Pb, Ca, Zn (In the increasing order of the reactivity)
Chapter:
Mg2+, Cu2+, Na1+, H1+ (In the order of preferential discharge at the cathode)
Chapter:
Li, K, Na, H (In the decreasing order of their ionization potential)
Chapter: [1] Periodic Table, Periodic Properties and Variations of Properties
F, B, N, O (In the increasing order of electron affinity)
Chapter: [1] Periodic Table, Periodic Properties and Variations of Properties
Ethane, methane, ethene, ethyne. (In the increasing order of the molecular weight)
Chapter: [12.1] Organic Compounds
Draw the electron dot structure of :
Nitrogen molecule [N = 7]
Chapter:
Draw the electron dot structure of :
Sodium chloride [Na = 11, Cl = 17]
Chapter:
The pH values of three solutions A, B and C are given in the table. Answer the folloiwng questions:
| Solution | pH value |
| A | 12 |
| B | 2 |
| C | 7 |
Which solution will have no effect on litmus solution?
Chapter: [3.2] Salts and their Preparations
The pH values of three solutions A, B and C are given in the table. Answer the folloiwng questions:
| Solution | pH value |
| A | 12 |
| B | 2 |
| C | 7 |
Which solution will liberate CO2 when reacted with sodium carbonate?
Chapter: [3.2] Salts and their Preparations
The pH values of three solutions A, B and C are given in the table. Answer the folloiwng
| Solution | pH value |
| A | 12 |
| B | 2 |
| C | 7 |
Which solution will turn red litmus solution blue?
Chapter:
Study the extract of the Periodic Table given below and answer the questions that follow. Give the alphabet corresponding to the element in question. DO NOT repeat an element. 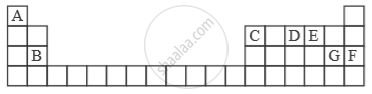
Which element forms electrovalent compound with G?
Chapter:
Study the extract of the Periodic Table given below and answer the questions that follow. Give the alphabet corresponding to the element in question. DO NOT repeat an element. 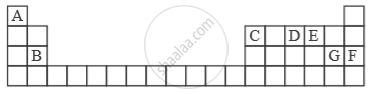
The ion of which element will migrate towards the cathode during electrolysis?
Chapter:
Study the extract of the Periodic Table given below and answer the questions that follow. Give the alphabet corresponding to the element in question. DO NOT repeat an element. 
Which non-metallic element has the valency of 2?
Chapter:
Study the extract of the Periodic Table given below and answer the questions that follow. Give the alphabet corresponding to the element in question. DO NOT repeat an element. 
Which is an inert gas?
Chapter:
Name the particles present in :
Strong electrolyte
Chapter: [3.2] Salts and their Preparations
Name the particles present in :
Weak electrolyte
Chapter: [3.1] Acids and Bases
Distinguish between the following pairs of compounds using the reagent given in the bracket.
Mangenese dioxide and copper (II) oxide. (using concentrated HCl)
Chapter: [8] Study of Compounds A. Hydrogen Chloride
Distinguish between the following pairs of compounds using the reagent given in the bracket.
Ferrous sulphate solution and ferric sulphate solution. (Using sodium hydroxide solution)
Chapter: [11] Study of Compounds D. Sulphuric Acid
Distinguish between the following pairs of compounds using the reagent given in the bracket.
Dilute hydrochloric acid and dilute sulphuric acid. (Using lead nitrate solution)
Chapter: [10] Study of Compounds C. Nitric Acid
Advertisements
Choose the method of preparation of the following salts, from the methods given in the list:
[List : A. Neutralization B. Precipitation C. Direct combination D. Substitution ]
(i) Lead chloride
(ii) Iron (II) sulphate
(iii) Sodium nitrate
(iv) Iron (III) chloride
Chapter: [3.2] Salts and their Preparations
Complete the following equations :
S + conc.HNO3 →
Chapter: [11] Study of Compounds D. Sulphuric Acid
Complete the following equations :
C + cons. H2SO4
Chapter: [11] Study of Compounds D. Sulphuric Acid
Complete the following equations :
S + conc.HNO3 →
Chapter: [11] Study of Compounds D. Sulphuric Acid
Write a balanced chemical equation for the preparation of :
Ethene from bromoethane
Chapter:
Write a balanced chemical equation for the preparation of :
Ethyne using calcium carbide
Chapter:
Write a balanced chemical equation for the preparation of :
Methane from sodium acetate
Chapter:
Give the chemical formula of :
Sodium aluminate
Chapter:
Answer the following questions based on the extraction of aluminium from alumina by Hall - Heroult’s Process :
What is the function of cryolite used along with alumina as the electrolyte?
Chapter:
Answer the following questions based on the extraction of aluminium from alumina by Hall - Heroult’s Process :
Why is powdered coke sprinkled on top of the electrolyte?
Chapter:
Answer the following questions based on the extraction of aluminium from alumina by Hall - Heroult’s Process :
Name the electrode, from which aluminium collected.
Chapter:
Match the alloys given in column I to the uses given in column II :
| Column I | Column II |
| (i) Duralumin | A. Electrical fuse |
| (ii) Solder | B. Surgical instruments |
| (iii) Brass | C. Aircraft body |
| (iv) Stainless Steel | D. Decorative articles |
Chapter: [7.1] Metals - Occurence
Identify the substance underlined:
The catalyst used to oxide ammonia.
Chapter: [9] Study of Compounds B. Ammonia
Identify the substance underlined:
The organic compound which when solidified, forms an ice like mass.
Chapter: [9] Study of Compounds B. Ammonia
Identify the substance underlined:
The dilute acid which is an oxidizing agent.
Chapter: [10] Study of Compounds C. Nitric Acid
Copper sulphate solution reacts with sodium hydroxide solution to form a precipitate of copper hydroxide according to the equation :
2NaOH + CuSO4 → Na2SO4 + Cu (OH)2 ↓
(i) What mass of copper hydroxide is precipitated by using 200 gm of sodium hydroxide?
[H = 1, O = 16, Na = 23, S = 32, Cu = 64]
(ii) What is the colour of the precipitate formed?
Chapter:
Find the empirical formula and the molecular formula of an organic compound from the data given below :
C = 75.92%, H = 6.32% and N = 17.76%
The vapour density of the compound is 39.5.
[C = 12, H = 1, N = 14]
Chapter: [5.3] Percentage Composition, Empirical and Molecular Formulae
Name the gas evolved in each of the following cases :
Alumina undergoes electrolytic reduction.
Chapter: [9] Study of Compounds B. Ammonia
Name the gas evolved in each of the following cases :
Ethene undergoes hydrogenation reaction.
Chapter: [9] Study of Compounds B. Ammonia
Name the gas evolved in each of the following cases :
Ammonia reacts with heated copper oxide.
Chapter: [9] Study of Compounds B. Ammonia
Study the flow chart given and give balanced equations to represent the reactions A, B and C:
\[\ce{Mg3N2 ->[A] NH3 <=>[B][C] NH4Cl}\]
Chapter: [9] Study of Compounds B. Ammonia
Copy and complete the following table which refers to the industrial method for the preparation of ammonia and sulphuric acid:
| Name of the compoun | Name of the process | Catalytic equation (with the catalyst) |
| Ammonia | (i)__________ | (ii) __________ |
| Sulhuric acid | (iii) ___________ | (iv) __________ |
Chapter: [9] Study of Compounds B. Ammonia
Other Solutions
Submit Question Paper
Help us maintain new question papers on Shaalaa.com, so we can continue to help studentsonly jpg, png and pdf files
CISCE previous year question papers ICSE Class 10 Chemistry with solutions 2018 - 2019
Previous year Question paper for CISCE ICSE Class 10 -2019 is solved by experts. Solved question papers gives you the chance to check yourself after your mock test.
By referring the question paper Solutions for Chemistry, you can scale your preparation level and work on your weak areas. It will also help the candidates in developing the time-management skills. Practice makes perfect, and there is no better way to practice than to attempt previous year question paper solutions of CISCE ICSE Class 10 .
How CISCE ICSE Class 10 Question Paper solutions Help Students ?
• Question paper solutions for Chemistry will helps students to prepare for exam.
• Question paper with answer will boost students confidence in exam time and also give you an idea About the important questions and topics to be prepared for the board exam.
• For finding solution of question papers no need to refer so multiple sources like textbook or guides.
