Advertisements
Advertisements
Question
Copy and complete the following table which refers to the industrial method for the preparation of ammonia and sulphuric acid:
| Name of the compoun | Name of the process | Catalytic equation (with the catalyst) |
| Ammonia | (i)__________ | (ii) __________ |
| Sulhuric acid | (iii) ___________ | (iv) __________ |
Advertisements
Solution
| Name of the compound | Name of the process | Catalytic equation (with the catalyst) |
| Ammonia | (i)Haber process | 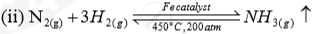 |
| Sulhuric acid | (iii)Contact process | 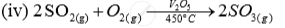 |
APPEARS IN
RELATED QUESTIONS
Name the gas evolved in each of the following cases :
Alumina undergoes electrolytic reduction.
Write an equation for the reaction to prepare one fertilizer from ammonia.
Give balanced reactions for the following conversion:
Ammonia to nitrogen using oxygen.
Give balanced reactions for the following conversion:
Ammonia solution to an amphoteric hydroxide.
Describe all what you will observe and write chemical equation, when limited amount of ammonia gas is passed through the following aqueous solution:
FeSO4
Write equation for the following:
Burning of ammonia in oxygen.
Write a balanced chemical equation for the lab preparation of ammonia.
The catalyst used in the oxidation of ammonia is ______.
