Advertisements
Advertisements
प्रश्न
Copy and complete the following table which refers to the industrial method for the preparation of ammonia and sulphuric acid:
| Name of the compoun | Name of the process | Catalytic equation (with the catalyst) |
| Ammonia | (i)__________ | (ii) __________ |
| Sulhuric acid | (iii) ___________ | (iv) __________ |
Advertisements
उत्तर
| Name of the compound | Name of the process | Catalytic equation (with the catalyst) |
| Ammonia | (i)Haber process | 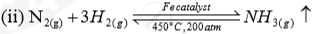 |
| Sulhuric acid | (iii)Contact process | 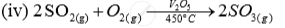 |
APPEARS IN
संबंधित प्रश्न
State one relevant observation for the following
When ammonia gas is burnt in an atmosphere of excess oxygen.
Complete the following equation.
4 NH3 +5O2 →
What important process starts with the reaction in above? Name the catalyst used.
Study the flow chart given and give balanced equations to represent the reactions A, B and C:
\[\ce{Mg3N2 ->[A] NH3 <=>[B][C] NH4Cl}\]
Name the following
Salt used to clean metal surface before soldering, tinning, etc.
Why is ammonium hydroxide used in qualitative analysis? Give two equations to justify your answer.
Give balanced equation of :
Action of heat an a mixture of copper an nitric acid
Give balanced reactions for the following conversion:
Ammonia to nitrogen using oxygen.
Describe all what you will observe and write chemical equation, when limited amount of ammonia gas is passed through the following aqueous solution:
FeSO4
State one relevant observation for the following:
Burning of ammonia in excess of oxygen.
