Advertisements
Advertisements
प्रश्न
Copy and complete the following table which refers to the industrial method for the preparation of ammonia and sulphuric acid:
| Name of the compoun | Name of the process | Catalytic equation (with the catalyst) |
| Ammonia | (i)__________ | (ii) __________ |
| Sulhuric acid | (iii) ___________ | (iv) __________ |
Advertisements
उत्तर
| Name of the compound | Name of the process | Catalytic equation (with the catalyst) |
| Ammonia | (i)Haber process | 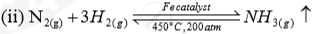 |
| Sulhuric acid | (iii)Contact process | 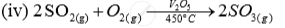 |
APPEARS IN
संबंधित प्रश्न
Give balanced chemical equations for Reaction of ammonia with excess chlorine.
Ammonia solution in water gives a blue precipitate when it combines with a solution of copper salt. The blue precipitate further dissolves in excess of an ammonia solution to give the azure blue solution. Explain with the equation.
Identify the substance underlined:
The catalyst used to oxide ammonia.
Name the following
Solution used to remove fat grease.
Name the following
An acidic gas which reacts with a basic gas liberating neutral gas.
Ammonia is manufactured by Haber Process.
Under what conditions do the reactants combine to form ammonia? Give a balanced equation for the reaction.
Write a balanced chemical equation for the following:
When an excess of ammonia is treated with chlorine.
Give balanced reactions for the following conversion:
Ammonia to nitrogen using trichloride.
Write equation for the following:
Catalytic oxidation of ammonia
Give a balanced chemical equation for the following:
Reaction of ammonia with excess of chlorine.
