English Medium
Academic Year: 2024-2025
Date & Time: 20th February 2025, 10:30 am
Duration: 3h
Advertisements
General Instructions:
Read the following instructions very carefully and strictly follow them:
- This question paper comprises 39 questions. All questions are compulsory.
- This question paper is divided into five sections - A, B, C, D and E.
- Section A - Question Nos. 1 to 20 are multiple choice type questions. Each question carries 1 mark.
- Section B - Question Nos. 21 to 26 are very short answer type questions. Each question carries 2 marks. Answer to these questions should be in the range of 30 to 50 words.
- Section C - Question Nos. 27 to 33 are short answer type questions. Each question carries 3 marks. Answer to these questions should in the range of 50 to 80 words.
- Section D - Question Nos. 34 to 36 are long answer type questions. Each question carries 5 marks. Answer to these questions should be in the range of 80 to 120 words.
- Section E - Question Nos. 37 to 39 are of 3 source-based/case-based units of assessment carrying 4 marks each with sub-parts.
- There is no overall choice. However, an internal choice has been provided in some sections. Only one of the alternatives has to be attempted in such questions.
A hydrocarbon which does not belong to the same homologous series of carbon compounds is ______.
C4H10
C6H14
C7H14
C10H22
Chapter:
The formula of washing soda is ______.
NaHCO3 . 6H2O
Na2CO3 . 6H2O
NaHCO3 . 10H2O
Na2CO3 . 10H2O
Chapter:
In the following experimental setup of electrolysis of water, if P and Q are the gases collected in the test tubes enclosing the electrodes R and S, then select the option/options in which the matching is correct:
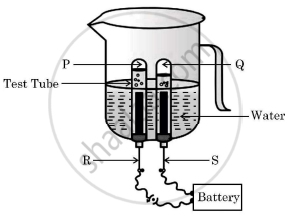
- P - Oxygen gas, R - Anode
- P - Hydrogen gas, R - Cathode
- Q - Hydrogen gas, S - Cathode
- Q - Oxygen gas, A - Anode
(i) and (ii)
(iii) and (iv)
(i) and (iii)
(ii) and (iv)
Chapter:
Select from the following a statement which is not true about burning of magnesium ribbon in air:
It burns with a dazzling white flame.
A white powder is formed on burning.
It is an endothermic reaction.
It is an example of a combination reaction.
Chapter:
The water of crystallization is present in:
- Bleaching Powder
- Plaster of Paris
- Washing Soda
- Baking Soda
(ii) and (iv)
(ii) and (iii)
(i) and (iii)
(i) and (iv)
Chapter:
The nature of an aqueous solution of potassium nitrate is ______.
acid
basic
neutral
alkaline
Chapter:
The colour of the solution observed after about 1 hour of placing iron nails in copper sulphate solution is ______.
Blue
Pale green
Yellow
Reddish brown
Chapter:
Mendel obtained the F2 generation by ______.
self-pollinating F1 generation plants.
cross-pollinating F1 generation plants with plants having dominant traits.
cross-pollinating F1 generation plants with plants having recessive traits.
cross-pollinating both the plants.
Chapter:
The basic filtration unit of the excretory system in human beings is ______.
Nephron
Urethra
Neuron
Urinary bladder
Chapter:
When a seed germinates, the root grows downwards and a small shoot grows upward. This shoot is known as ______.
radicle
stem
cotyledon
plumule
Chapter:
A pair of endocrine glands located in the human brain is ______.
Parathyroid and Pituitary
Pineal and Thymus
Hypothalamus and Thymus
Hypothalamus and Pineal
Chapter:
Identify from the following the ray diagram which shows the correct path of the reflected ray for the ray incident on a concave mirror, as shown:


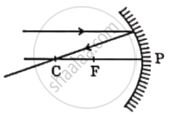
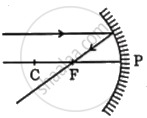
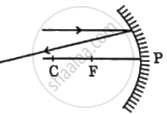
Chapter:
In the human alimentary canal, the digestive juices secreted by the gastric glands are ______.
Bile, Trypsin, Pepsin
Hydrochloric acid, Pepsin, Mucus
Lipase, Bile, Mucus
Salivary amylase, Pepsin, Bile
Chapter:
Most of the refraction for the light rays entering the eye occurs at ______.
Iris
Pupil
Crystalline lens
Outer surface of cornea
Chapter:
The incorrect statement about ozone is:
It is a deadly poisonous gas.
It shields the surface of the earth from UV radiation from the sun.
It is used as a refrigerant and in fire extinguishers.
It is formed by combining an oxygen molecule with a free oxygen atom.
Chapter:
Green plants occupy the first trophic level in every food chain because they ______.
exist over a large area.
have very less concentration of harmful chemicals.
have to feed large number of herbivores.
can synthesize food by photosynthesis.
Chapter:
Assertion (A): Xylem tissue moves water and minerals obtained from the soil by the roots.
Reason (R): Xylem tissue is found only in the roots of a plant.
Both Assertion (A) and Reason (R) are true and Reason (R) is the correct explanation of Assertion (A).
Both Assertion (A) and Reason (R) are true and Reason (R) is not the correct explanation of Assertion (A).
Assertion (A) is true, but Reason (R) is false.
Assertion (A) is false, but Reason (R) is true.
Chapter:
Assertion (A): Food web is a network of several food chains operating in an ecosystem.
Reason (R): Food web decreases the stability of an ecosystem.
Both Assertion (A) and Reason (R) are true and Reason (R) is the correct explanation of Assertion (A).
Both Assertion (A) and Reason (R) are true and Reason (R) is not the correct explanation of Assertion (A).
Assertion (A) is true, but Reason (R) is false.
Assertion (A) is false, but Reason (R) is true.
Chapter:
Assertion (A): Carbon and its compounds are our major sources of fuels.
Reason (R): Most of the carbon compounds on burning release a large amount of heat and light.
Both Assertion (A) and Reason (R) are true and Reason (R) is the correct explanation of Assertion (A).
Both Assertion (A) and Reason (R) are true and Reason (R) is not the correct explanation of Assertion (A).
Assertion (A) is true, but Reason (R) is false.
Assertion (A) is false, but Reason (R) is true.
Chapter:
Assertion (A): In the common domestic circuits the earth wire is connected to a metallic plate buried deep inside the earth.
Reason (R): Earth wire ensures that any leakage of current to the metallic body of the appliance keeps its potential to that of the earth, so the user may not get a severe electric shock.
Both Assertion (A) and Reason (R) are true and Reason (R) is the correct explanation of Assertion (A).
Both Assertion (A) and Reason (R) are true and Reason (R) is not the correct explanation of Assertion (A).
Assertion (A) is true, but Reason (R) is false.
Assertion (A) is false, but Reason (R) is true.
Chapter:
Cinnabar is an ore of a metal ‘X’. When this ore is heated in air, it is first converted into oxide of ‘X’ (XO) and then reduced to metal ‘X’ on further heating.
Identify metal ‘X’ and write chemical equations for the reactions that occur in the above processes.
Chapter:
“Proteins control the expression of various characters.” Explain this statement by taking the example of “tallness” as a characteristic in plants.
Chapter:
Explain the mechanism of inheritance used by sexually reproducing organisms to ensure the stability of DNA of the species.
Chapter:
Advertisements
A doctor finds in one of his patients that he is not maintaining a proper posture and balance of his body. State the region of brain and also the part of brain which is responsible for it.
Chapter:
Give reasons:
The sky appears dark to passengers flying at very high altitudes.
Chapter:
Give a scientific reason:
Danger signal lights are red in colour.
Chapter:
What is a rainbow?
Chapter: [10] The Human Eye and the Colourful World
Why do we see a rainbow in the sky only after rainfall?
Chapter: [10] The Human Eye and the Colourful World
Study the figure in which the path of a ray of light going from Medium 1 to Medium 2 is shown.
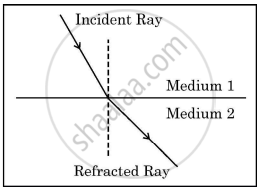
- Out of the two Media - Medium 1 and Medium 2, in which is the speed of light more?
- State reason of bending of the refracted ray away from the normal.
- Express refractive index of Medium 2 with respect to Medium 1 in terms of speed of light in two media.
Chapter:
Indicate the flow of energy in a food chain operating in an ecosystem. Why is it uni-directional? Give reason.
Chapter:
The gene combination of purple flowered pea plants is denoted as (WW) and that of white flowered pea plants as (ww), when these two plants are crossed F1 generation is obtained.
- List two observations made by Mendel in F1 generation plants.
- Give the (i) percentage of white flowered plants and (ii) ratio of the gene combinations WW, Ww and ww in F2 generation.
- Write one difference between dominant and recessive trait.
Chapter:
In an experiment a student dipped pH papers in four different solutions A, B, C and D and reported his observations as given below:
| Solution | A | B | C | D |
| Colour of pH paper | Light green | Blue | Green | Red |
- In which one of these solutions is the concentration of
- hydrogen/hydronium ions;
- hydroxyl ions maximum?
- Give one example each of the two solutions identified in (i) above.
- What would be the pH of the resultant mixture obtained when these two solutions are mixed together in equal proportions? Justify your answer.
Chapter:
Name the gas liberated when an acid reacts with a metal. How is this gas tested?
Chapter:
Write the chemical equation for the reaction of zinc metal with hydrochloric acid. Name the compound of zinc obtained in the given case.
Chapter:
Write the chemical equation for the reaction of zinc metal with sodium hydroxide. Name the compound of zinc obtained in the given case.
Chapter:
An object of size 5 cm is placed at a distance of 30 cm from the optical centre of a converging lens of focal length 20 cm. Use lens formula to determine the position and size of the image formed.
Chapter:
Write a correctly balanced equation for the following:
When steam is passed over red hot iron.
Chapter:
Write a balanced chemical equation for the reaction that occurs when natural gas is burnt in air.
Chapter:
Write a balanced chemical equation for the reaction that occurs when glucose reacts with oxygen in the cells of our body and provides energy.
Chapter:
Explain the statement ‘the potential difference between two points is 1 volt’.
Chapter:
What does the symbol given below represent in an electric circuit? Write one function.
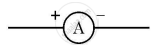
Chapter:
What does the symbol given below represent in an electric circuit? Write one function.
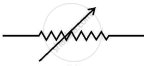
Chapter:
A student placed a candle flame at different distances from a convex lens and focused its image on a screen. He recorded his observation in tabular form as given below:
| S. No. | Distance of flame from the lens (cm) |
Distance of the image from the lens |
| 1 | −90 | +18 |
| 2 | −60 | +20 |
| 3 | −40 | +24 |
| 4 | −30 | +30 |
| 5 | −24 | +40 |
| 6 | −20 | +60 |
| 7 | −18 | +90 |
| 8 | −12 | +120 |
Analyse the observation table and on the basis of your analysis only, answer the following questions (without doing any calculations):
- What is the focal length of the convex lens used? Give reason to justify your answer.
- Which one of the sets of observations is not correct and why?
- Draw a ray diagram to show image formation for any correct set of observations.
Chapter:
Draw a ray diagram to show the dispersion of white light.
Chapter: [10] The Human Eye and the Colourful World
What is spectrum? Give a reason for its formation.
Chapter:
Advertisements
What changes are seen in boys at the time of puberty?
Chapter:
Why are testes in human males located outside the abdominal cavity in the scrotum?
Chapter:
List any three techniques of contraception used by humans. Which one of these is not meant for males?
Chapter:
Name the part performing the following function in the human female reproductive system:
Production of eggs
Chapter:
Name the part performing the following function in the human female reproductive system:
Site of fertillization
Chapter:
Name the part performing the following function in the human female reproductive system:
Site of implantation
Chapter:
Name the part performing the following function in the human female reproductive system:
Entry of the sperms
Chapter:
What changes are observed in the uterus subsequent to the implantation of a young embryo?
Chapter:
What changes are observed in the uterus if an egg does not get fertilized?
Chapter:
Name an alcohol and a carboxylic acid having two carbon atoms in their structures. Draw their structures and state how this alcohol can be converted into a carboxylic acid. What happens when these two compounds react in the presence of an acid? Write chemical equations for the reactions involved in the two cases mentioned above.
Chapter:
Why are soaps not considered suitable for washing clothes in a region where water is hard? How is this problem overcome?
Chapter:
Study the following electric circuit diagram and answer the questions that follow:
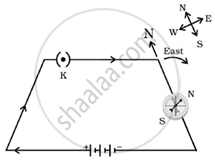
- What does the circuit diagram show?
- What will happen if the direction of current is reversed? Justify your answer, giving a circuit diagram.
Chapter:
Name and state the rule to determine the direction of the magnetic field associated with a straight current-carrying conductor.
Chapter:
Draw the pattern of magnetic field lines of a bar magnet.
Chapter:
Draw the pattern of magnetic field lines of a current-carrying solenoid.
Chapter:
List two distinguishing features between the magnetic fields produced by a bar magnet and a solenoid.
Chapter:
Study the following three diagrams in which the entry of an electron in a magnetic field is shown. Identify the case in which the magnetic force experienced by the electron is (i) maximum, and (ii) minimum. Give reason for your answers in each case.
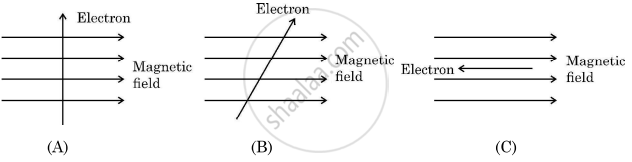
Chapter:
| Many pure metals like copper, iron and gold are very soft and as such are considered unsuitable for certain uses. Metallic objects around us such as cooking utensils, statues, ornaments, guns, etc., are actually not made up of pure metals. Instead of pure metals, alloys are used in the design of most of the useful objects. Making alloys enhances the basic properties of a metal which is the primary constituent (metal) of an alloy. |
- How does electrical conductivity and melting point of a metal change when it is converted to its alloy by mixing a small amount of an element in it? (1)
- Name an alloy used for welding two wires together in an electric circuit. Write its major constituents. (1)
-
- What are alloys? How is ‘Brass’ (an alloy) prepared? (2)
OR - What is stainless steel? How is it prepared? Write one important property which makes it more useful in making cooking utensils as compared to its primary metal. (2)
- What are alloys? How is ‘Brass’ (an alloy) prepared? (2)
Chapter:
|
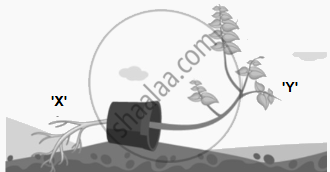
The growth movements of plant parts in which the direction of the stimulus determines the direction of the response are known as tropic movements or tropism. Plants also have non-directional movements, which may not be growth-dependent.
|
- Name the movement which causes ‘X’ and ‘Y’ to grow downwards and upwards respectively. (1)
- Write the name of a hormone that plays a major role in
- falling of leaves.
- rapid cell division. (1)
-
- Leaves of the sensitive plant move very quickly in response to ‘touch’. How is this stimulus of touch communicated and explain how the movement takes place? (2)
OR - Name the plant hormone which is synthesized at the shoot tip. How does this hormone help the plant to bend towards light? (2)
- Leaves of the sensitive plant move very quickly in response to ‘touch’. How is this stimulus of touch communicated and explain how the movement takes place? (2)
Chapter:
Study the circuit shown in which two resistors X and Y of resistances 3 Ω and 6 Ω respectively are joined in series with a battery of 2 V.

- Draw a circuit diagram showing the above two resistors X and Y joined in parallel with the same battery and the same ammeter and voltmeter. (1)
- In which combination of resistors will the (i) potential difference across X and Y and (ii) current through X and Y, be the same? (1)
-
- Find the current drawn from the battery by the series combination of the two resistors (X and Y). (2)
OR - Determine the equivalent resistance of the parallel combination of the two resistors (X and Y). (2)
- Find the current drawn from the battery by the series combination of the two resistors (X and Y). (2)
Chapter:
Other Solutions
Submit Question Paper
Help us maintain new question papers on Shaalaa.com, so we can continue to help studentsonly jpg, png and pdf files
CBSE previous year question papers Class 10 Science with solutions 2024 - 2025
Previous year Question paper for CBSE Class 10 Science-2025 is solved by experts. Solved question papers gives you the chance to check yourself after your mock test.
By referring the question paper Solutions for Science, you can scale your preparation level and work on your weak areas. It will also help the candidates in developing the time-management skills. Practice makes perfect, and there is no better way to practice than to attempt previous year question paper solutions of CBSE Class 10.
How CBSE Class 10 Question Paper solutions Help Students ?
• Question paper solutions for Science will helps students to prepare for exam.
• Question paper with answer will boost students confidence in exam time and also give you an idea About the important questions and topics to be prepared for the board exam.
• For finding solution of question papers no need to refer so multiple sources like textbook or guides.