Advertisements
Advertisements
Question
Write names and formulae of hydrocarbons containing a single and a double bond (one example for each). Give one characteristic chemical property of each.
Advertisements
Solution
Methane is a hydrocarbon which has a single bond. Its formula is CH4.
The characteristic chemical property of methane is the substitution reaction. It is a reaction in which one or more hydrogen atoms are replaced by some other atoms.
Chemical equation:
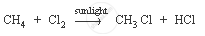
Ethene is a hydrocarbon containing a double bond. Its formula is C2H4.
The characteristic chemical property of ethene is the addition reaction. It is a reaction in which unsaturated compound (ethene) is combined with another substance to form a single product (saturated compound).
Chemical equation:
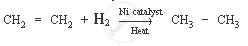
APPEARS IN
RELATED QUESTIONS
Explain the action of phosphorous trichloride with ethanol. Write the balanced chemical equation of the above reaction.
Write the name and structural formula of the compound obtained when ethanol is heated at 443 K with excess of conc. H2SO4. Also write chemical equation for the reaction stating the role of conc. H2SO4 in it.
Fill in the following blank with suitable word:
The next homologue of C2H5OH is ...............
Match the formulae in group A with appropriate names from group B:
Group A: CH3COOH, CH3CHO, CH3OH
Group B: Ethanol, Methanol, Ethanal, Ethanoic acid
Give one exampleof the compound having the following functional group:
Alcohol group
Give the harmful effects of drinking alcohol.
Consider the following organic compound:
CH3OH, C2H5OH, CH3COCH3, CH3COOH, C2H5COOH, C4H9COOC2H5, CH4, C2H6, CH3CHO, HCHO
Out of these compound:
Which compound is used to sterilise wounds and syringes?
How is the spurious alcohol obtained?
Write the rnolecular formula and structural forrnula of ethanol.
Ethane, Ethene, Ethanoic acid, Ethyne, Ethanol
From the compounds given above, name
The compound with -OH as apart of its structure.
Write a balanced chemical equation for the following:
Ethanol under high pressure and low temperature is treated with acidified potassium dichoromate.
Convert ethanol into ethene using hot conc. H2SO4. Give only a balanced equation.
What are the functional groups? Name any two compounds containing functional groups.
3 mL of ethanol is taken in a test tube and warmed gently in a water bath. A 5% solution of alkaline potassium permanganate is added to the first drop by drop to this solution, then in excess.
- How is a 5% solution of KMnO4 prepared?
- State the role of alkaline potassium permanganate in this reaction. What happens on adding it in excess?
- Write the chemical equation of this reaction.
How is ethanoic acid prepared from ethanol? Give the chemical equation.
Write the chemical equation for the following:
Oxidation of ethanol
Draw electron dot structure of Ethanol.
Give the balanced chemical equation of the following reaction:
Oxidation of ethanol by acidified potassium dichromate.
Give the balanced chemical equation of the following reaction:
Oxidation of ethanol by acidified potassium dichromate.
