Advertisements
Advertisements
प्रश्न
Write names and formulae of hydrocarbons containing a single and a double bond (one example for each). Give one characteristic chemical property of each.
Advertisements
उत्तर
Methane is a hydrocarbon which has a single bond. Its formula is CH4.
The characteristic chemical property of methane is the substitution reaction. It is a reaction in which one or more hydrogen atoms are replaced by some other atoms.
Chemical equation:
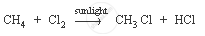
Ethene is a hydrocarbon containing a double bond. Its formula is C2H4.
The characteristic chemical property of ethene is the addition reaction. It is a reaction in which unsaturated compound (ethene) is combined with another substance to form a single product (saturated compound).
Chemical equation:
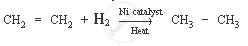
APPEARS IN
संबंधित प्रश्न
Give the structural formula of ethanol.
The compound formed when ethene reacts with Hydrogen is ________. (CH4, C2H6, C3H8)
Organic compounds having functional group are known as
functional group are known as
How would you test for an alcohol?
Consider the following organic compound:
CH3OH, C2H5OH, CH3COCH3, CH3COOH, C2H5COOH, C4H9COOC2H5, CH4, C2H6, CH3CHO, HCHO
Out of these compound:
Which compound is used to sterilise wounds and syringes?
Identify the functional group of the CH3OH ?
From the given list, name the compound with –COOH as part of its structure.
From the given list, name the homologue of the homologous series with the general formula CnH2n.
Write the equation for the following lab preparation:
Methanol from iodomethane.
Find the odd one out and explain:
State how the following conversion can be carried out:
Ethyl alcohol to ethene.
Choose the correct alternative and rewrite the following:
To observe the hydro and clearly, Raju should see it first under the low power lens and then under the high power lens in order to see _____________
Generally _______ is called spirit.
Which of the following are used as anesthetics?
100% pure ethanol is called ______.
Give the balanced chemical equation of the following reaction:
Oxidation of ethanol by acidified potassium dichromate.
______ react with dil. HNO3 to evolve hydrogen gas.
Give the balanced chemical equation of the following reaction:
Combustion of ethanol.
Give the balanced chemical equation of the following reaction:
Combustion of ethanol.
Give the balanced chemical equation of the following reaction:
Oxidation of ethanol by acidified potassium dichromate.
