Advertisements
Advertisements
प्रश्न
Write names and formulae of hydrocarbons containing a single and a double bond (one example for each). Give one characteristic chemical property of each.
Advertisements
उत्तर
Methane is a hydrocarbon which has a single bond. Its formula is CH4.
The characteristic chemical property of methane is the substitution reaction. It is a reaction in which one or more hydrogen atoms are replaced by some other atoms.
Chemical equation:
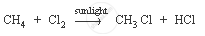
Ethene is a hydrocarbon containing a double bond. Its formula is C2H4.
The characteristic chemical property of ethene is the addition reaction. It is a reaction in which unsaturated compound (ethene) is combined with another substance to form a single product (saturated compound).
Chemical equation:
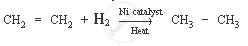
APPEARS IN
संबंधित प्रश्न
Write a balanced chemical equations for Heating of Ethanol at 170°C in the presence of conc. Sulphuric acid
Fill in the following blank with suitable word:
The next higher homologue of ethane is ...............
Write the name of the following functional group:

How would you test for an alcohol?
How do the boiling point and melting point change in the homologous series of alcohols?
How is the spurious alcohol obtained?
Vinegar is prepared by the bacterial oxidation of ______.
Write a chemical reaction to show the dehydration of ethanol.
How is methane gas prepared in laboratory?
Give the necessary conditions and equations of getting ethanol from:
(a) Alkyl halide
(b) An ethene
Ethanol can be converted to ethene which can be changed to ethane.
Choose the correct word or phrase from the options given below to complete the following sentences:
Converting ethanol to ethene requires the use of_________(Concentrated hydrochloric acid, concentrated nitric cid, concentrated sulphuric acid).
Write balanced equations to show the preparation of the following:
Ethene from ethanol
Write the structural formulae of the following compound:
Ethane
Answer the following question.
Write the chemical formula and name of the compound which is the active ingredient of all alcoholic drinks. List its two uses.
Write the chemical equation and name of the product formed when this compound reacts with -
(i) Sodium metal
(ii) hot concentrated sulphuric acid
Ethanol is also known as which of these?
Ethene is formed when ethanol at 443 K is heated with excess of concentrated sulphuric acid. What is the role of sulphuric acid in this reaction? Write the balanced chemical equation of this reaction.
How would you bring about the following conversions? Name the process and write the reaction involved.
What happens when ethanol is heated at 443K in the presence of conc. H2SO4? Write the role of conc. H2SO4 in this case.
