Advertisements
Advertisements
Question
Write IUPAC name of the following compound:
\[\begin{array}{cc}
\ce{HO - CH2 - CH - CH2 - OH}\\
|\phantom{..}\\
\ce{OH}
\end{array}\]
Advertisements
Solution
Propane-1, 2, 3-triol
APPEARS IN
RELATED QUESTIONS
Name the following compound according to IUPAC system.

Write IUPAC name of the following compound:
\[\begin{array}{cc}
\ce{H3C - CH - CH2 - CH - CH - CH2 - CH3}\\
\phantom{}|\phantom{.............}|\phantom{......}|\phantom{.........}\\
\phantom{}\ce{OH}\phantom{..........}\ce{OH}\phantom{...}\ce{C2H5}\phantom{......}
\end{array}\]
Write structures of the compounds whose IUPAC names are as follows:
3-Chloromethylpentan-1-ol.
Ethylidene dichloride when boiled with aqueous solution of NaOH yields _______.
(A) formaldehyde
(B) acetaldehyde
(C) acetone
(D) ethyl methyl ketone
Write IUPAC name of the following compound (CH3)2 N − CH2CH3
Write the IUPAC name of the following compound:

Write the structures of the products when Butan-2-ol reacts with SOCl2
In the dehydration of alcohols to alkenes by heating with concentrated sulphuric acid, the initiation step is:
(1) formation of carbonation
(2) formation of an ester
(3) protonation of the alcohol molecule
(4) elimination of water
Write structural formula for pentane-1,4-diol.
An example of a compound with functional group – O – is ____________.
Cresol has ____________.
n-Propyl alcohol and isopropyl alcohol can be chemically distinguished by which reagent?
IUPAC name of m-cresol is ____________.
The major product formed by the reaction:
\[\begin{array}{cc}
\ce{CH3CH-CH2Br ->[CH3O^-][CH3OH] is}\\
|\phantom{................}\\
\ce{CH3}\phantom{.............}
\end{array}\]
\[\ce{Phenol ->[Zn, dust] 'X' ->[CH3Cl][Anhy. AlCl3] 'Y' ->[Alkaline][KMnO4] 'Z'}\]
The product ‘Z’ is:
Write the IUPAC name of the compound given below.
\[\begin{array}{cc}
\phantom{}\ce{CH3 - CH2 - C = C - OH}\\
\phantom{........}|\phantom{....}|\phantom{}\\
\phantom{..............}\ce{CH3 CH2OH}
\end{array}\]
Assertion: p-nitrophenol is more acidic than phenol.
Reason: Nitro group helps in the stabilisation of the phenoxide ion by dispersal of negative charge due to resonance.
Explain why Lewis acid is not required in bromination of phenol?
Write structural formulae for:
p-Nitrophenol
The IUPAC name of 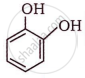 is ______.
is ______.
