Advertisements
Advertisements
Question
Write IUPAC name of the following compound:
\[\begin{array}{cc}
\ce{CH3 - CH - CH - CH3}\\
|\phantom{......}|\phantom{..}\\
\ce{OH}\phantom{...}\ce{OH}\phantom{}
\end{array}\]
Advertisements
Solution
Butane-2, 3-diol
APPEARS IN
RELATED QUESTIONS
Write the IUPAC name of the given compound:

Write IUPAC name of the following compound:
C6H5 – O – C7H15(n−)
Ethylidene dichloride when boiled with aqueous solution of NaOH yields _______.
(A) formaldehyde
(B) acetaldehyde
(C) acetone
(D) ethyl methyl ketone
Natalite is a mixture of
(a) diethyl ether and methanol
(b) diethyl ether and ethanol
(c) dimethyl ether and methanol
(d) dimethyl ether and ethanol
How is phenol converted into the following?
picric acid
Write structural formulae for 3-Methoxyhexane
Write IUPAC name of the following

C6H5OCH2CH3 is called:
Cresol has ____________.
The major product formed by the reaction:
\[\begin{array}{cc}
\ce{CH3CH-CH2Br ->[CH3O^-][CH3OH] is}\\
|\phantom{................}\\
\ce{CH3}\phantom{.............}
\end{array}\]
The product of acid catalysed hydration of 2-phenylpropene is:
The correct acidic strength order of the following is:

(I)
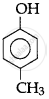
(II)

(III)
Assertion: p-nitrophenol is more acidic than phenol.
Reason: Nitro group helps in the stabilisation of the phenoxide ion by dispersal of negative charge due to resonance.
Assertion: Like bromination of benzene, bromination of phenol is also carried out in the presence of Lewis acid.
Reason: Lewis acid polarises the bromine molecule.
Convert the following:
Ethyl alcohol into ethyl acetate.
Identify A and B in the following:

Draw structure of the following compound.
2-Methoxypropane
Write structural formulae for:
Salicylic acid
The IUPAC name of 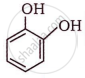 is ______.
is ______.
