Advertisements
Advertisements
Question
Write IUPAC name of the following compound:
\[\begin{array}{cc}
\phantom{................}\ce{CH3}\\
\phantom{.............}|\\
\ce{CH3 - CH - CH - C - CH3}\\
|\phantom{......}|\phantom{......}|\\
\phantom{...}\ce{CH3\phantom{.}}\phantom{..}\ce{OH}\phantom{...}\ce{CH3}
\end{array}\]
Advertisements
Solution
2, 2, 4-Trimethylpentan-3-ol
APPEARS IN
RELATED QUESTIONS
Name the following compound according to IUPAC system.

Name the following compound according to IUPAC system.
\[\begin{array}{cc}
\ce{H2C = CH - CH - CH2 - CH2 - CH3}\\
|\phantom{..........}\\
\ce{OH}\phantom{........}
\end{array}\]
Write IUPAC name of the following compound:
\[\begin{array}{cc}
\ce{CH3 - O - CH2 - CH - CH3}\\
\phantom{..........}|\\
\phantom{............}\ce{CH3}
\end{array}\]
Give IUPAC name of the following ether:
CH3OCH2CH2Cl
Natalite is a mixture of
(a) diethyl ether and methanol
(b) diethyl ether and ethanol
(c) dimethyl ether and methanol
(d) dimethyl ether and ethanol
3-Methylbutane-2-ol on heating with HI gives ______
How is phenol converted into the following?
picric acid
Give reasons Fluoride ion has higher hydration enthalpy than chloride ion.
Write the structures of the products when Butan-2-ol reacts with SOCl2
Write structural formulae for Cyclohex-2-en-1-ol.
One of the following is not a dihydroxy derivative of benzene.
Butane-2-ol is ____________.
IUPAC name of m-cresol is ____________.
The correct acidic strength order of the following is:

(I)
(II)

(III)
Arrange the following compounds in decreasing order of acidity.
\[\ce{H2O, ROH, HC ≡ CH}\]
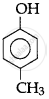
Match the structures of the compounds given in Column I with the name of the compounds given in Column II.
| Column I | Column II | |
| (i) |  |
(a) Hydroquinone |
| (ii) |  |
(b) Phenetole |
| (iii) | 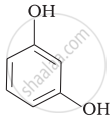 |
(c) Catechol |
| (iv) |  |
(d) o-Cresol |
| (v) |  |
(e) guinone |
| (vi) | 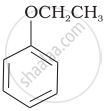 |
(f) Resorcinol |
| (g) Anisole |
Convert the following:
Ethyl alcohol into ethyl acetate.
Draw structure of the following compound.
2. 5-Diethylphenol
Write structural formulae for:
p-Nitrophenol
