Advertisements
Advertisements
Question
Which of the following is the correct expression for the equation of state of van der Waals gas?
Options
`["P" + "a"/("n"^2"V"^2)]("V" - "nb") = "nRT"`
`["P" + "na"/("n"^2"V"^2)]("V" - "nb") = "nRT"`
`["P" + "an"^2/("V"^2)]("V" - "nb") = "nRT"`
`["P" + ("n"^2"a"^2)/("V"^2)]("V" - "nb") = "nRT"`
Advertisements
Solution
`["P" + "an"^2/("V"^2)]("V" - "nb") = "nRT"`
APPEARS IN
RELATED QUESTIONS
The value of the universal gas constant depends upon
In what way real gases differ from ideal gases.
Which of the following gases would you expect to deviate from ideal behavior under conditions of low-temperature F2, Cl2, or Br2? Explain.
A plot of volume (V) versus temperature (T) for a gas at constant pressure is a straight line passing through the origin. The plots at different values of pressure are shown in Figure. Which of the following order of pressure is correct for this gas?
Under which of the following two conditions applied together, a gas deviates most from the ideal behaviour?
(i) Low pressure
(ii) High pressure
(iii) Low temperature
(iv) High temperature
Compressibility factor, Z, of a gas is given as Z = `(pV)/(nRT)`. What is the value of Z for an ideal gas?
Compressibility factor, Z, of a gas is given as Z = `(pV)/(nRT)`. For real gas what will be the effect on value of Z above Boyle’s temperature?
Assertion (A): At constant temperature, pV vs V plot for real gases is not a straight line.
Reason (R): At high pressure all gases have \[\ce{Z}\] > 1 but at intermediate pressure most gases have \[\ce{Z}\] < 1.
Isotherms of carbon dioxide gas are shown in figure. Mark a path for changing gas into liquid such that only one phase (i.e., either a gas or a liquid) exists at any time during the change. Explain how the temperature, volume and pressure should be changed to carry out the change.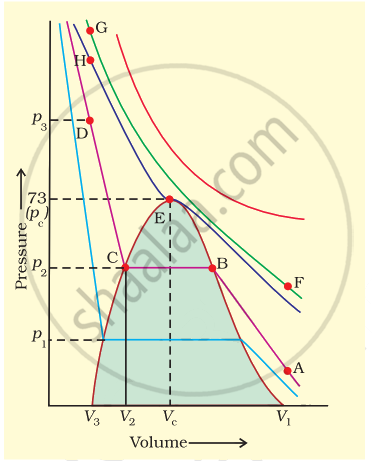
Choose the correct option for the total pressure (in atm.) in a mixture of 4g \[\ce{O2}\] and 2g \[\ce{H2}\] confined in a total volume of one litre at 0°C is ______.
[Given R = 0.082 L atm mol−1K−1, T = 273 K]
