Advertisements
Advertisements
प्रश्न
Which of the following is the correct expression for the equation of state of van der Waals gas?
विकल्प
`["P" + "a"/("n"^2"V"^2)]("V" - "nb") = "nRT"`
`["P" + "na"/("n"^2"V"^2)]("V" - "nb") = "nRT"`
`["P" + "an"^2/("V"^2)]("V" - "nb") = "nRT"`
`["P" + ("n"^2"a"^2)/("V"^2)]("V" - "nb") = "nRT"`
Advertisements
उत्तर
`["P" + "an"^2/("V"^2)]("V" - "nb") = "nRT"`
APPEARS IN
संबंधित प्रश्न
Can a Van der Waals gas with a = 0 be liquefied? explain.
Suppose there is a tiny sticky area on the wall of a container of gas. Molecules hitting this area stick there permanently. Is the pressure greater or less than on the ordinary area of walls?
Explain whether a gas approaches ideal behavior or deviates from ideal behaviour if it is compressed to a smaller volume at a constant temperature.
Explain whether a gas approaches ideal behavior or deviates from ideal behaviour if the temperature is raised while keeping the volume constant.
Explain whether a gas approaches ideal behavior or deviates from ideal behaviour if more gas is introduced into the same volume and at the same temperature.
Which of the following gases would you expect to deviate from ideal behavior under conditions of low-temperature F2, Cl2, or Br2? Explain.
If 1 gram of each of the following gases are taken at STP, which of the gases will occupy (a) greatest volume and (b) smallest volume?
\[\ce{CO, H2O, CH4 , NO}\]
Compressibility factor, Z, of a gas is given as Z = `(pV)/(nRT)`. For real gas what will be the effect on value of Z above Boyle’s temperature?
Match the following graphs of ideal gas with their co-ordinates:
| Graphical representation | x and y co-ordinates |
(i) 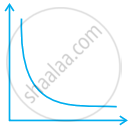 |
(a) pV vs. V |
(ii) 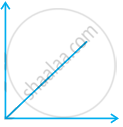 |
(b) p vs. V |
(iii) 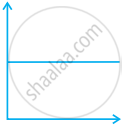 |
(c) p vs. `1/V` |
In van der Waal's equation for the real gas, the expression for the net force of attraction amongst the gas molecules is given by:
