Advertisements
Advertisements
Questions
What is the difference between molality and molarity?
Differentiate between molarity and molality.
Advertisements
Solution
| Sr. No. | Molarity | Molality |
| 1. | Molarity is the concentration of a material determined as the number of moles of solute dissolved in one litre of solution. | Molality is the concentration calculated as the number of moles of solute found in one kilogram of solvent. |
| 2. | Molarity is denoted by the symbol M. | Molality is denoted by the symbol m. |
| 3. | The molarity formula is moles per litre. | The molality formula is moles per kilogram. |
| 4. | Molarity is impacted by temperature changes. | Molality is not affected by temperature changes. |
| 5. | Changes in pressure affect molarity. | Pressure changes do not affect molality. |
| 6. | Molarity can lead to an imprecise and inaccurate concentration. | Molality can lead to an accurate and precise concentration measurement. |
APPEARS IN
RELATED QUESTIONS
Calculate the amount of carbon dioxide that could be produced when 2 moles of carbon are burnt in 16 g of dioxygen.
What is the concentration of sugar (C12H22O11) in mol L–1 if its 20 g are dissolved in enough water to make a final volume up to 2 L?
Solve the following problem:
Write the following number in ordinary decimal form:
3.49 × 10−11
Solve the following problem:
Write the following number in ordinary decimal form:
3.75 × 10−1
Solve the following problem:
Write the following number in ordinary decimal form:
0.011 × 10−3
Solve the following problem:
Perform the following calculation. Round off your answer to two digits.
`33/(9.00xx10^-4)`
Solve the following problem:
Perform the following calculation. Round off your answer to two digits.
`(1.4xx10^9)/((2.77xx10^3)(3.76xx10^5))`
Solve the following problem:
What weight of calcium oxide will be formed on heating 19.3 g of calcium carbonate?
(At. wt.: Ca = 40; C = 12; O = 16)
Give an example of each mixture having the following characteristics. Suggest a suitable method to separate the components of this mixture
Two immiscible liquids.
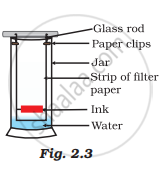
A child wanted to separate the mixture of dyes constituting a sample of ink. He marked a line by the ink on the filter paper and placed the filter paper in a glass containing water as shown in Fig.2.3. The filter paper was removed when the water moved near the top of the filter paper.
(i) What would you expect to see, if the ink contains three different coloured components?
(ii) Name the technique used by the child.
(iii) Suggest one more application of this technique.
If 500 mL of a 5 M solution is diluted to 1500 mL, what will be the molarity of the solution obtained?
Sulphuric acid reacts with sodium hydroxide as follows:
\[\ce{H2SO4 + 2NaOH -> Na2SO4 + 2H2O}\]
When 1 L of 0.1 M sulphuric acid solution is allowed to react with 1 L of 0.1 M sodium hydroxide solution, the amount of sodium sulphate formed and its molarity in the solution obtained is:
(i) 0.1 mol L–1
(ii) 7.10 g
(iii) 0.025 mol L–1
(iv) 3.55 g
Which of the following terms are unitless?
(i) Molality
(ii) Molarity
(iii) Mole fraction
(iv) Mass percent
Match the following physical quantities with units
| Physical quantity | Unit |
| (i) Molarity | (a) g mL–1 |
| (ii) Mole fraction | (b) mol |
| (iii) Mole | (c) Pascal |
| (iv) Molality | (d) Unitless |
| (v) Pressure | (e) mol L–1 |
| (vi) Luminous intensity | (f) Candela |
| (vii) Density | (g) mol kg–1 |
| (viii) Mass | (h) Nm–1 |
| (i) kg |
The molarity of pure water is ______.
An aqueous KCl solution of density 1.20 g mL-1 has a molality of 3.30 mol kg-1. The molarity of the solution in mol L-1 is ______. (Nearest integer)
The molarity of urea (molar mass 60 g mol−1) solution by dissolving 15 g of urea in 500 cm3 of water is ______.
