Advertisements
Advertisements
Question
What happens when 2-Nitropropane boiled with HCl?
Advertisements
Solution
\[\begin{array}{cc}
\ce{H}\phantom{...........................................}\\
|\phantom{...........................................}\\
\ce{CH3 - C - NO2 ->[HCl/H2O] CH3 - C = O + \underset{(Nitrots oxide)}{N2O} + H2O}\\
\phantom{}|\phantom{.........................}|\phantom{..................}\\
\phantom{}\ce{CH3}\phantom{....................}\ce{\underset{(Acetone)}{CH3}}\phantom{..............}
\end{array}\]
APPEARS IN
RELATED QUESTIONS
Which of the following reagent can be used to convert nitrobenzene to aniline?
Which one of the following will not undergo Hofmann bromamide reaction?
When 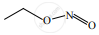 is reduced with Sn/HCl the pair of compounds formed are ____________.
is reduced with Sn/HCl the pair of compounds formed are ____________.
What happens when oxidation of tert-butylamine with KMnO4?
What happens when oxidation of acetone oxime with trifluoroperoxy acetic acid
How will you convert nitrobenzene into hydrozobenzene?
How will you convert nitrobenzene into aniline?
Identify A to E in the following sequence of reactions.

Account for the following.
Aniline does not undergo Friedel-Crafts reaction.
Account for the following.
Aniline does not undergo Friedel – Crafts reaction.
