Advertisements
Advertisements
Question
How will you convert nitrobenzene into hydrozobenzene?
Advertisements
Solution
Conversion of nitrobenzene into hydrazobenzene:
\[\ce{\underset{(nitrobenzene)}{2C6H5NO2} ->[Zn/NaOH] [C6H5 - N = N - C6H5] ->[2(H)] \underset{(Hydrozobenzene)}{C6H5 - NH - NH - C6H5}}\]
APPEARS IN
RELATED QUESTIONS
Which one of the following will not undergo Hofmann bromamide reaction?
Which one of the following nitro compounds does not react with nitrous acid?
Which of the following reaction is not correct.
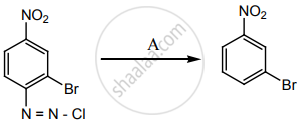
‘A’ is:
What happens when 2-Nitropropane boiled with HCl?
Arrange the following.
In increasing order of basic strength aniline, p-toluidine and p-nitro aniline
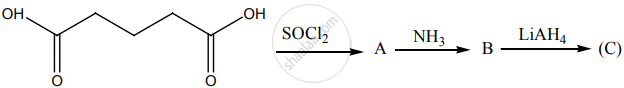
Identify A, B and C.
Account for the following.
Aniline does not undergo Friedel – Crafts reaction
Account for the following.
Aniline does not undergo Friedel-Crafts reaction.
Account for the following.
Aniline does not undergo Friedel – Crafts reaction.
