Advertisements
Advertisements
Question
How will you convert nitrobenzene into N-phenylhydroxylamine?
Advertisements
Solution
Conversion of nitrobenzene into N-phenylhydroxylamine:
\[\ce{\underset{(nitrobenzene)}{C6H5NO2} ->[Zn/NH4Cl][H2O; \Delta] \underset{(N-phenylhydroxylamine)}{C6H5 - NHOH} + ZnO}\]
APPEARS IN
RELATED QUESTIONS
\[\ce{CH3CH2Br ->[aq NaOH][\Delta] A ->[KMnO4/H^+][\Delta] B ->[NH3][\Delta] C ->[Br2/NaOH] D}\] ‘D’ is:
Which one of the following nitro compounds does not react with nitrous acid?
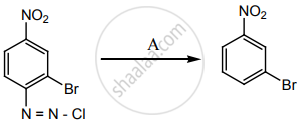
‘A’ is:
Nitrobenzene on reaction with at 80-100°C forms which one of the following products?
Secondary nitro alkanes react with nitrous acid to form ____________.
When 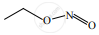 is reduced with Sn/HCl the pair of compounds formed are ____________.
is reduced with Sn/HCl the pair of compounds formed are ____________.
How will you convert nitrobenzene into 1, 3, 5-trinitrobenzene?
How will you convert nitrobenzene into hydrozobenzene?
Account for the following.
Aniline does not undergo Friedel–Crafts reaction
Account for the following:
Aniline does not undergo Friedel – Crafts reaction.
