Advertisements
Advertisements
Question
Arrange the following.
In increasing order of basic strength aniline, p-toluidine and p-nitro aniline
Advertisements
Solution
The electron-donating groups increase the basic strength of amines while the electron-withdrawing groups decrease the basic strength of amines. Therefore p-nitro aniline is the weakest base followed by aniline while p-toluidine, which has methyl group and therefore it is the strongest base.
Basic strength increases in the order:
P-nitro aniline < aniline < p-toluidine
APPEARS IN
RELATED QUESTIONS
\[\ce{CH3CH2Br ->[aq NaOH][\Delta] A ->[KMnO4/H^+][\Delta] B ->[NH3][\Delta] C ->[Br2/NaOH] D}\] ‘D’ is:
Nitrobenzene on reaction with at 80-100°C forms which one of the following products?
How will you convert nitrobenzene into azoxybenzene?
Identify compounds A, B and C in the following sequence of reaction.
\[\ce{C6H5NH2 ->[(CH3CO2)O][pyridine] A ->[HNO3][H2SO4, 288 K] B ->[H2O/H^+] C}\]
Identify A, B and C.
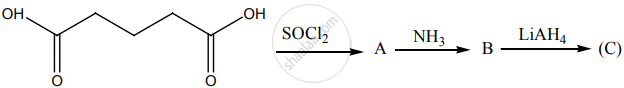
Account for the following
Aniline does not undergo Friedel–Crafts reaction
Account for the following.
Aniline does not undergo Friedel–Crafts reaction.
Account for the following.
Aniline does not undergo Friedel – Crafts reaction.
Account for the following:
Aniline does not undergo Friedel – Crafts reaction.
Account for the following.
Aniline does not undergo Friedel–Crafts reaction
