Advertisements
Advertisements
प्रश्न
Arrange the following.
In increasing order of basic strength aniline, p-toluidine and p-nitro aniline
Advertisements
उत्तर
The electron-donating groups increase the basic strength of amines while the electron-withdrawing groups decrease the basic strength of amines. Therefore p-nitro aniline is the weakest base followed by aniline while p-toluidine, which has methyl group and therefore it is the strongest base.
Basic strength increases in the order:
P-nitro aniline < aniline < p-toluidine
APPEARS IN
संबंधित प्रश्न
There are two isomers with the formula CH3NO2. How will you distinguish between them?
What happens when 2-Nitropropane boiled with HCl?
How will you convert nitrobenzene into hydrozobenzene?
How will you convert nitrobenzene into N-phenylhydroxylamine?
How will you convert nitrobenzene into aniline?
Identify compounds A, B and C in the following sequence of reaction.
\[\ce{C6H5NH2 ->[(CH3CO2)O][pyridine] A ->[HNO3][H2SO4, 288 K] B ->[H2O/H^+] C}\]
Write a short note on the following.
Hofmann’s bromide reaction
Identify A, B and C.
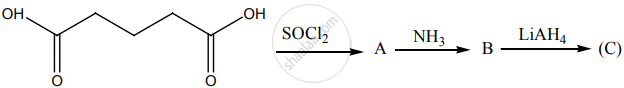
Account for the following
Aniline does not undergo Friedel–Crafts reaction
Account for the following.
Aniline does not undergo Friedel – Crafts reaction.
