Advertisements
Advertisements
प्रश्न
What happens when oxidation of acetone oxime with trifluoroperoxy acetic acid
Advertisements
उत्तर
Oxidation of acetone oxime with trifluoroperoxy acetic acid gives 2-nitropropane.
\[\begin{array}{cc}
\ce{CH3 - C = N - OH ->[CF3COOH][(O)] CH3 - CH - NO2}\\
\phantom{}|\phantom{.............................}|\phantom{..}\\
\phantom{.}\ce{\underset{(Acetone oxime)}{CH3}}\phantom{..................}\ce{\underset{(2-nitropropane)}{CH3}}\phantom{}
\end{array}\]
APPEARS IN
संबंधित प्रश्न
The method by which aniline cannot be prepared is ____________.
Which one of the following nitro compounds does not react with nitrous acid?
Nitrobenzene on reaction with at 80-100°C forms which one of the following products?
When 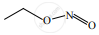 is reduced with Sn/HCl the pair of compounds formed are ____________.
is reduced with Sn/HCl the pair of compounds formed are ____________.
Write down the possible isomers of the C4H9NO2 give their IUPAC names.
How will you convert nitrobenzene into azoxybenzene?
How will you convert nitrobenzene into N-phenylhydroxylamine?
How will you convert nitrobenzene into aniline?
Account for the following:
Aniline does not undergo Friedel – Crafts reaction.
Account for the following.
Aniline does not undergo Friedel – Crafts reaction.
