Advertisements
Advertisements
Question
The method by which aniline cannot be prepared is ____________.
Options
degradation of benzamide with Br2/NaOH
potassium salt of phthalimide treated with chlorobenzene followed by hydrolysis with aqueous NaOH solution
reduction of Nitrobenzene with LiAlH4
reduction of nitrobenzene by Sn/HCl
Advertisements
Solution
The method by which aniline cannot be prepared is potassium salt of phthalimide treated with chlorobenzene followed by hydrolysis with aqueous NaOH solution.
APPEARS IN
RELATED QUESTIONS
Which of the following reagent can be used to convert nitrobenzene to aniline?
Which one of the following will not undergo Hofmann bromamide reaction?
Which one of the following nitro compounds does not react with nitrous acid?
When 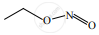 is reduced with Sn/HCl the pair of compounds formed are ____________.
is reduced with Sn/HCl the pair of compounds formed are ____________.
What happens when 2-Nitropropane boiled with HCl?
What happens when Nitrobenzene undergoes electrolytic reduction in a strongly acidic medium?
Identify compounds A, B and C in the following sequence of reaction.
\[\ce{C6H5NO2 ->[Fe/HCl] A ->[HNO2][273 K] B ->[C6H5OH] C}\]
Identify A to E in the following sequence of reactions.

Account for the following
Aniline does not undergo Friedel–Crafts reaction
Account for the following.
Aniline does not undergo Friedel – Crafts reaction.
