Advertisements
Advertisements
Question
What happens when oxidation of tert-butylamine with KMnO4?
Advertisements
Solution
In general, primary amines, in which the –NH2 group is attached to a tertiary carbon can be oxidised with KMnO4 to the corresponding nitro compound in excellent yield.
Therefore 3°-butylamine oxidised to give 2-methyl-2-nitropropane.
\[\begin{array}{cc}
\phantom{...}\ce{CH3}\phantom{...................}\ce{CH3}\phantom{}\\
\phantom{}|\phantom{.......................}|\phantom{}\\
\ce{CH3 - C - NH2 ->[KMnO4] CH3 - C - NO2}\\
\phantom{}|\phantom{.......................}|\phantom{}\\
\phantom{........}\ce{\underset{(3°-butylamine)}{CH3}}\phantom{........}\ce{\underset{(2-methyl-2-nitropropane)}{CH3}}\phantom{..}
\end{array}\]
APPEARS IN
RELATED QUESTIONS
Which of the following reagent can be used to convert nitrobenzene to aniline?
\[\ce{CH3CH2Br ->[aq NaOH][\Delta] A ->[KMnO4/H^+][\Delta] B ->[NH3][\Delta] C ->[Br2/NaOH] D}\] ‘D’ is:
Which of the following reaction is not correct.
What happens when 2-Nitropropane boiled with HCl?
What happens when oxidation of acetone oxime with trifluoroperoxy acetic acid
How will you convert nitrobenzene into aniline?
Identify compounds A, B and C in the following sequence of reaction.
\[\ce{C6H5NO2 ->[Fe/HCl] A ->[HNO2][273 K] B ->[C6H5OH] C}\]
Identify A, B and C.
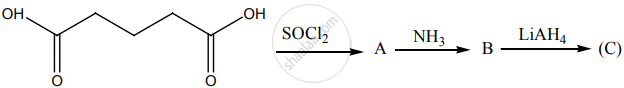
Account for the following.
Aniline does not undergo Friedel – Crafts reaction.
Account for the following.
Aniline does not undergo Friedel – Crafts reaction.
