Advertisements
Advertisements
Question
What is the action of following reagents on glucose?
bromine water
Advertisements
Solution
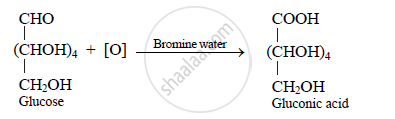
Action of bromine water on glucose: The oxidation of glucose with bromine water
(which is a mild oxidizing agent) forms gluconic acid. This indicates presence of
aldehyde group.
APPEARS IN
RELATED QUESTIONS
Describe laboratory method for preparation of glucose.
What is the action of following reagents on glucose?
dilute HNO3
What is the action of following reagents on glucose?
hydroxyl amine
Stachyose is an example of _______.
(A) monosaccharides
(B) disaccharides
(C) trisaccharides
(D) tetrasaccharides
Write the formula of the complex copper (II) hexacyanoferrate (II).
Which component of starch is a branched polymer of α-glucose and insoluble in water?
Hydrolysis of starch yields ____________.
Which among the following is the simplest sugar?
Which of the following statements is not true about glucose?
Carbohydrates are classified on the basis of their behaviour on hydrolysis and also as reducing or non-reducing sugar. Sucrose is a:
(i) monosaccharide
(ii) disaccharide
(iii) reducing sugar
(iv) non-reducing sugar
In nucleoside a base is attached at 1′ position of sugar moiety. Nucleotide is formed by linking of phosphoric acid unit to the sugar unit of nucleoside. At which position of sugar unit is the phosphoric acid linked in a nucleoside to give a nucleotide?
Under what conditions glucose is converted to gluconic and saccharic acid?
Which sugar is called invert sugar? Why is it called so?
During curdling of milk, what happens to sugar present in it?
How do you explain the presence of five – OH groups in glucose molecule?
In the acetylation of glucose, which group is involved in the reaction
A nucleoside is made of:-
Which of the following base is not present in RNA?
A nucleoside acids, the sequence is
Among the naturally occuring carbohydrates, furanose ring is found in the:
When sucrose is heated with cone. HNO3 the product is ______.
How is glucose prepared on commercial scale?
