Advertisements
Advertisements
Question
Use Bohr’s model of hydrogen atom to obtain the relationship between the angular momentum and the magnetic moment of the revolving electron.
Advertisements
Solution
In the Bohr model of the hydrogen atom, the electron is modeled as a point negative charge rotating in a circular orbit about a fixed axis about a nucleus.
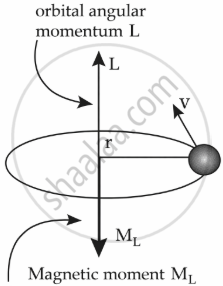
Let us consider,
r = radius of the orbit
v = velocity
e = charge of electron
m = mass of electron
Time period (T) = `"circumference"/"velocity"`
= `(2 pi r)/v`
Current (I) = `(-e)/T`
= `(-e)/((2 pi r)/v)`
= `(-e v)/(2 pi r)`
The magnetic moment due to a current loop enclosing an area A is given by:
ML = IA
= `(-e V)/(2 pi r) xx A`
= `(-e v)/(2 pi r) xx pi r^2`
= `(-e r v)/2`
= `(-m e r v)/(2 m)`
L = Angular momentum = mvr
So, ML = `(-e)/(2 m) L`
RELATED QUESTIONS
State Bohr’s third postulate for hydrogen (H2) atom. Derive Bohr’s formula for the wave number. Obtain expressions for longest and shortest wavelength of spectral lines in ultraviolet region for hydrogen atom
State Bohr’s postulate of hydrogen atom which successfully explains the emission lines in the spectrum of hydrogen atom. Use Rydberg formula to determine the wavelength of Hα line. [Given: Rydberg constant R = 1.03 × 107 m−1]
In Bohr’s model of the hydrogen atom, the radius of the first orbit of an electron is r0 . Then, the radius of the third orbit is:
a) `r_0/9`
b) `r_0`
c) `3r_0`
d) `9r_0`
Which of the following parameters are the same for all hydrogen-like atoms and ions in their ground states?
Find the wavelength of the radiation emitted by hydrogen in the transitions (a) n = 3 to n= 2, (b) n = 5 to n = 4 and (c) n = 10 to n = 9.
Given below are two statements:
Statements I: According to Bohr's model of an atom, qualitatively the magnitude of velocity of electron increases with decrease in positive charges on the nucleus as there is no strong hold on the electron by the nucleus.
Statement II: According to Bohr's model of an atom, qualitatively the magnitude of velocity of electron increase with a decrease in principal quantum number.
In light of the above statements, choose the most appropriate answer from the options given below:
The value of angular momentum for He+ ion in the first Bohr orbit is ______.
What is the energy of an electron in stationary state corresponding to n = 2?
Calculate the radius of the second orbit of He+.
Which from following is CORREСТ relationship between wavelength and momentum of electron?
