Advertisements
Advertisements
प्रश्न
Use Bohr’s model of hydrogen atom to obtain the relationship between the angular momentum and the magnetic moment of the revolving electron.
Advertisements
उत्तर
In the Bohr model of the hydrogen atom, the electron is modeled as a point negative charge rotating in a circular orbit about a fixed axis about a nucleus.
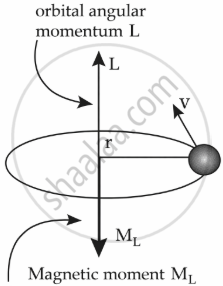
Let us consider,
r = radius of the orbit
v = velocity
e = charge of electron
m = mass of electron
Time period (T) = `"circumference"/"velocity"`
= `(2 pi r)/v`
Current (I) = `(-e)/T`
= `(-e)/((2 pi r)/v)`
= `(-e v)/(2 pi r)`
The magnetic moment due to a current loop enclosing an area A is given by:
ML = IA
= `(-e V)/(2 pi r) xx A`
= `(-e v)/(2 pi r) xx pi r^2`
= `(-e r v)/2`
= `(-m e r v)/(2 m)`
L = Angular momentum = mvr
So, ML = `(-e)/(2 m) L`
संबंधित प्रश्न
Obtain an expression for the radius of Bohr orbit for H-atom.
Calculate the radius of Bohr’s fifth orbit for hydrogen atom
Draw a neat, labelled energy level diagram for H atom showing the transitions. Explain the series of spectral lines for H atom, whose fixed inner orbit numbers are 3 and 4 respectively.
The gravitational attraction between electron and proton in a hydrogen atom is weaker than the Coulomb attraction by a factor of about 10−40. An alternative way of looking at this fact is to estimate the radius of the first Bohr orbit of a hydrogen atom if the electron and proton were bound by gravitational attraction. You will find the answer interesting.
The electron in hydrogen atom is initially in the third excited state. What is the maximum number of spectral lines which can be emitted when it finally moves to the ground state?
The numerical value of ionization energy in eV equals the ionization potential in volts. Does the equality hold if these quantities are measured in some other units?
Why was a change in the Bohr Model of atom required? Due to which important development (s), concept of movement of an electron in an orbit was replaced by, the concept of probability of finding electron in an orbital? What is the name given to the changed model of atom?
In form of Rydberg's constant R, the wave no of this first Ballmer line is
Taking the Bohr radius as a0 = 53 pm, the radius of Li++ ion in its ground state, on the basis of Bohr’s model, will be about ______.
When an electron falls from a higher energy to a lower energy level, the difference in the energies appears in the form of electromagnetic radiation. Why cannot it be emitted as other forms of energy?
