Advertisements
Advertisements
प्रश्न
Use Bohr’s model of hydrogen atom to obtain the relationship between the angular momentum and the magnetic moment of the revolving electron.
Advertisements
उत्तर
In the Bohr model of the hydrogen atom, the electron is modeled as a point negative charge rotating in a circular orbit about a fixed axis about a nucleus.
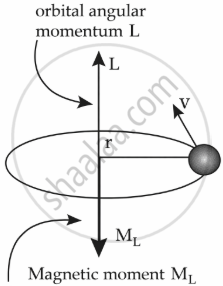
Let us consider,
r = radius of the orbit
v = velocity
e = charge of electron
m = mass of electron
Time period (T) = `"circumference"/"velocity"`
= `(2 pi r)/v`
Current (I) = `(-e)/T`
= `(-e)/((2 pi r)/v)`
= `(-e v)/(2 pi r)`
The magnetic moment due to a current loop enclosing an area A is given by:
ML = IA
= `(-e V)/(2 pi r) xx A`
= `(-e v)/(2 pi r) xx pi r^2`
= `(-e r v)/2`
= `(-m e r v)/(2 m)`
L = Angular momentum = mvr
So, ML = `(-e)/(2 m) L`
संबंधित प्रश्न
Calculate the energy required for the process
\[\ce{He^+_{(g)} -> He^{2+}_{(g)} + e^-}\]
The ionization energy for the H atom in the ground state is 2.18 ×10–18 J atom–1
Suppose, the electron in a hydrogen atom makes transition from n = 3 to n = 2 in 10−8 s. The order of the torque acting on the electron in this period, using the relation between torque and angular momentum as discussed in the chapter on rotational mechanics is
When a photon stimulates the emission of another photon, the two photons have
(a) same energy
(b) same direction
(c) same phase
(d) same wavelength
According to Bohr’s theory, the angular momentum of an electron in 5th orbit is ______.
Ratio of longest to shortest wavelength in Balmer series is ______.
According to the Bohr theory of H-atom, the speed of the electron, its energy and the radius of its orbit varies with the principal quantum number n, respectively, as:
The energy of an electron in hth orbit of hydrogen atom is –13.6/n2ev energy required to excite the electron from the first orbit to the third orbit is
Given below are two statements:
Statements I: According to Bohr's model of an atom, qualitatively the magnitude of velocity of electron increases with decrease in positive charges on the nucleus as there is no strong hold on the electron by the nucleus.
Statement II: According to Bohr's model of an atom, qualitatively the magnitude of velocity of electron increase with a decrease in principal quantum number.
In light of the above statements, choose the most appropriate answer from the options given below:
Hydrogen atom from excited state comes to the ground state by emitting a photon of wavelength λ. If R is the Rydberg constant then the principal quantum number n of the excited state is ______.
The energy of an electron in the nth orbit of the hydrogen atom is En = -13.6/n2eV. The negative sign of energy indicates that ______.
