Advertisements
Advertisements
Question
The phenomenon in which compounds having different structural formulae have the same molecular formula is called _______.
Options
structural isomerism
catenation
homologous
functional group
Advertisements
Solution
The phenomenon in which compounds having different structural formulae have the same molecular formula is called structural isomerism.
APPEARS IN
RELATED QUESTIONS
Write the next homologue of the following: C4H6
What is meant by homologous series of carbon compounds?
Write the name and formula of the 2nd member of homologous series having general formula CnH2n.
Write the name and formula of the 2nd member of homologous series having general formula CnH2n – 2.
Write the molecular formula of two consecutive members of homologous series of aldehydes. State which part of these compounds determines their
- physical and
- chemical properties
Write the molecular formula of the 2nd and 3rd member of the homologous series where the first member is ethyne.
Write the names and formulae for the first three members of the homologous series for chloroalkanes.
The molecular formula of the third member of the homologous series of ketones is:
(a) C4H8O
(b) C3H6O
(c) C5H10O
(d) C6H12O
What is the difference in the molecular formula of any two adjacent homologues in terms of molecular mass?
Why homologous series of carbon compounds are so called? Write chemical formula of two consecutive members of a homologous series and state the part of these compounds that determines their (i) physical properties, and (ii) chemical properties.
Distinguish between homologous organs and analogous organs. In which category would you place wings of a bird and wings of a bat? Justify your answer giving a suitable reason.
The general formula of Alkane is _________________
Assertion (A): In a homologous series of alcohols, the formula for the second member is C2H5OH and the third member is C3H7OH.
Reason (R): The difference between the molecular masses of the two consecutive members of a homologous series is 144.
The general molecular formula for the homologous series of alkynes is _______.
Observe the structural formula and answer the following questions.
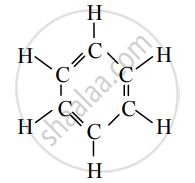
- Write the name of the given hydrocarbon.
- The given hydrocarbon is included in which type of hydrocarbon?
- What is the kind of compounds with the above characteristic structure called?
Which of the following does not belong to the same homologous series?
Consider the carbon compounds having following molecular formula:
(i) C3H6 (ii) C3H8 (iii) C4H6 (iv) C6H6 (v) C6H12
- State the number of double covalent bonds present in C3H6.
- Write the formula of first member of the homologous series to which the carbon compound C4H6 belongs.
- Which one of the above compounds forms a ring structure of carbon atoms?
- Identify, which of the above compounds, is a member of alkane series.
Consider the carbon compounds having following molecular formula:
(i) C2H2 (ii) C2H5 (iii) C3H7OH (iv) C2H6COOH (v) CH3CHO
- Identify which one of the above compounds, is a member of aldehyde series.
- Write the general formula of the series to which compound C2H2 belongs.
- Which one of the above compounds has triple bonds between carbon-carbon atoms?
- Write the molecular formula of the first member of the homologous series to which the compound C3H7OH belongs.
