Advertisements
Advertisements
Question
Choose the correct option and rewrite the statement.
The number of electrons in the outermost shell of alkali metals is _______.
Options
1
2
3
7
Advertisements
Solution
The number of electrons in the outermost shell of alkali metals is 1.
APPEARS IN
RELATED QUESTIONS
An element P (atomic number 20) reacts with an element Q (atomic number 17) to form a compound. Answer the following questions giving reason:
Write the position of P and Q in the Modern Periodic Table and the molecular formula of the compound formed when P reacts with Q.
The element which forms a basis oxide has the atomic number of:
(a) 18
(b) 17
(c) 14
(d) 19
An element X combines with oxygen to form an oxide XO. This oxide is electrically conducting.
(a) How many electrons would be there in the outermost shell of the element X?
(b) To which group of the periodic tables does the element X belong?
(c) Write the formula of the compound formed when X reacts with chlorine.
A non-metal X which is the largest constituent of air combines with hydrogen when heated in the presence of iron as catalyst to form a gas Y. When gas Y is treated with sulphuric acid, it forms a compound Z which is used as a chemical fertiliser.
(a) What are X, Y and Z?
(b) To which group of periodic table does X belong?
(c) Name the periodic table in which X is placed.
(d) Which element is placed just before X in the period?
(e) Which element is placed just after X in the period?
Alkaline earth metals have valency 2. This means that their position in the modern periodic table is in ______.
What is the need for classification of elements?
Elements belonging to the same group have the same valency.
If the distance between two Cl atoms in a Cl2 molecule is 1.98Å, then the radius of the Cl atom is ______.
Among the given species A–, A+, and A, the smallest one in size is ______.
An element X from group 2 of the Periodic Table reacts with Y from group 17 to form a compound. Give the formula of the compound.
The elements of group 16 are called ______
In which group are inert elements placed?
The element with atomic number 26 will be found in group ______
Arrange the following elements in the order of their decreasing metallic character
Na, Si, Cl, Mg, Al
What type of oxide would Eka– aluminium form?
Identify and name the metals out of the following elements whose electronic configurations are given below.
- 2, 8, 2
- 2, 8, 1
- 2, 8, 7
- 2, 1
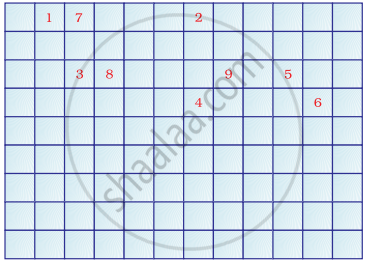
Complete the following cross word puzzle
Across:
(1) An element with atomic number 12.
(3) Metal used in making cans and member of Group 14.
(4) A lustrous non-metal which has 7 electrons in its outermost shell.
Down:
(2) Highly reactive and soft metal which imparts yellow colour when subjected to flame and is kept in kerosene.
(5) The first element of second Period
(6) An element which is used in making fluorescent bulbs and is second member of Group 18 in the Modern Periodic Table
(7) A radioactive element which is the last member of halogen family.
(8) Metal which is an important constituent of steel and forms rust when exposed to moist air.
(9) The first metalloid in Modern Periodic Table whose fibres are used in making bullet-proof vests
Complete the following triads by inserting the missing elements.
Cl, ______, I
How are elements grouped into various families in the periodic table?
Identify the following:
An element in Period 1 which can be placed in both Group 1 and Group 17 of the Periodic Table.
