Advertisements
Advertisements
प्रश्न
Choose the correct option and rewrite the statement.
The number of electrons in the outermost shell of alkali metals is _______.
पर्याय
1
2
3
7
Advertisements
उत्तर
The number of electrons in the outermost shell of alkali metals is 1.
APPEARS IN
संबंधित प्रश्न
An element P (atomic number 20) reacts with an element Q (atomic number 17) to form a compound. Answer the following questions giving reason:
Write the position of P and Q in the Modern Periodic Table and the molecular formula of the compound formed when P reacts with Q.
State whether the following statement is true of false:
The elements in a group have consecutive atomic numbers.
An element A from group 14 of the periodic table combines with an element B from group 16.
(i) What type of chemical bond is formed?
(ii) Give the formula of the compound formed.
Find the neutral atom in the periodic table which has the same number of electrons as K+ and Cl−. What is this number?
The molecular formula of the chloride of element X is XCl. This compound is a solid having a high melting point. Which of the following elements be present in the same group as X.
What was the basis of the earliest attempts made for classification and grouping of elements?
Write the name.
The atom having smallest atomic radius from group 1.
The basis of the classifications proposed by Dobereiner, Newlands and Mendeleev was ______.
The number of periods and groups in the periodic table are ______
Moseley’s periodic table is based on atomic mass.
Identify the bond between H and F in the HF molecules.
An atom of an element has the electronic configuration 2,8,2. To which group does it belong?
An element X from group 2 of the Periodic Table reacts with Y from group 17 to form a compound. Give the formula of the compound.
Consider the following elements 20Ca, 8Or 18Ar, 16S, 4Be, 2He
Which of the above elements would you expect to be in group 16 of the Periodic Table?
The elements of group 16 are called ______
Element ‘A’ has electronic configuration 2, 7; ‘B’ has electronic configuration 2, 8, 5 and ‘C’ has electronic configuration 2, 8, 7. Which two elements will exhibit similar chemical properties?
An element with the atomic number 40 is placed in the period.
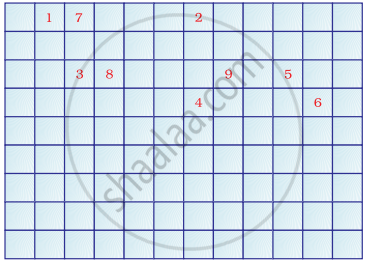
Complete the following cross word puzzle
Across:
(1) An element with atomic number 12.
(3) Metal used in making cans and member of Group 14.
(4) A lustrous non-metal which has 7 electrons in its outermost shell.
Down:
(2) Highly reactive and soft metal which imparts yellow colour when subjected to flame and is kept in kerosene.
(5) The first element of second Period
(6) An element which is used in making fluorescent bulbs and is second member of Group 18 in the Modern Periodic Table
(7) A radioactive element which is the last member of halogen family.
(8) Metal which is an important constituent of steel and forms rust when exposed to moist air.
(9) The first metalloid in Modern Periodic Table whose fibres are used in making bullet-proof vests
Relate the names of the following scientists with the statements given below.
Arranged elements into groups containing three elements each.
Complete the following triads by inserting the missing elements.
Ca, ______, Ba
