Advertisements
Advertisements
प्रश्न
The molecular formula of the chloride of element X is XCl. This compound is a solid having a high melting point. Which of the following elements be present in the same group as X.
पर्याय
Na
Mg
Al
Si
Advertisements
उत्तर
Molecular formula of the chloride of an element X is XCl. This compound is a solid having high melting point. An element to be present in the same group as X is Na.
APPEARS IN
संबंधित प्रश्न
An element P (atomic number 20) reacts with an element Q (atomic number 17) to form a compound. Answer the following questions giving reason:
Write the position of P and Q in the Modern Periodic Table and the molecular formula of the compound formed when P reacts with Q.
Write the electronic configuration of two elements X and Y whose atomic numbers are 20 and 17 respectively. Write the molecular formula of the compound formed when element X reacts with element Y. Draw the electron-dot structure of the product and also state the nature of the bond formed between both the elements.
As element X has mass number 40 and contains 21 neutrons in its atom. To which group of the periodic table does it belong?
The following diagram shows a part of the periodic table containing first three periods in which five elements have been represented by the letters a, b, c, d and e (which are not their chemical symbols):
-
1 18 a 2 13 14 15 16 17 b c d e
(i) Select the letter which represents an alkali metal.
(ii) Select the letter which represents a nobles gas.
(iii) Select the letter which represents a halogen.
(iv) What type of bond is formed between a and e?
(v) What type of bond is formed between d and e?
Find the neutral atom in the periodic table which has the same number of electrons as K+ and Cl−. What is this number?
Atoms of eight elements A, B, C, D, E, F, G and H have the same number of electron shells but different number of electrons in their outermost shells. It was found that elements A and G combine to form an ionic compound. This ionic compound is added in a small amount to almost all vegetables and dishes during cooking. Oxides of elements A and B are basis in nature while those of elements E and F acidic. The oxide of element D is, however, almost neutral. Based on the above information, answer the following questions:
(a) To which group or period of the periodic table do these elements belong?
(b) What would be the nature of compound formed by a combination of elements B and F?
(c) Which two of these elements could definitely be metals?
(d) Which one of the eight elements is most likely to be found in gaseous state at room temperature?
(e) If the number of electrons in the outermost shell of elements C and G be 3 and 7 respectively, write formula of the compound formed by the combination of C and G.
An element X (atomic number 17) reacts with an element Y (atomic number 20) to form a compound.
(a) Write the position of these elements in the modern periodic table.
(b) Write the formula of the compound formed.
Justify your answer in each case.
Alkaline earth metals have valency 2. This means that their position in the modern periodic table is in ______.
Example for liquid metal is ______.
Match the following
| 1. | Galvanisation | Noble gas elements |
| 2. | Calcination | Coating with Zn |
| 3. | Redox reaction | Silver-tin amalgam |
| 4. | Dental filling | Alumino thermic process |
| 5. | Group 18 elements | Heating in the absence of air |
What property forms the basis of identification?
Which of the following hydroxides is most basic?
An element is placed in 2nd Group and 3rd Period of the Periodic Table, burns in presence of oxygen to form a basic oxide.
- Identify the element
- Write the electronic configuration
- Write the balanced equation when it burns in the presence of air
- Write a balanced equation when this oxide is dissolved in water
- Draw the electron dot structure for the formation of this oxide
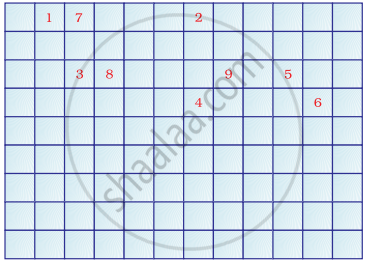
Complete the following cross word puzzle
Across:
(1) An element with atomic number 12.
(3) Metal used in making cans and member of Group 14.
(4) A lustrous non-metal which has 7 electrons in its outermost shell.
Down:
(2) Highly reactive and soft metal which imparts yellow colour when subjected to flame and is kept in kerosene.
(5) The first element of second Period
(6) An element which is used in making fluorescent bulbs and is second member of Group 18 in the Modern Periodic Table
(7) A radioactive element which is the last member of halogen family.
(8) Metal which is an important constituent of steel and forms rust when exposed to moist air.
(9) The first metalloid in Modern Periodic Table whose fibres are used in making bullet-proof vests
Relate the names of the following scientists with the statements given below.
Arranged elements in a group of seven with increasing atomic masses and eighth elements with similar properties kept below the first like eight note in an act of music.
Complete the following triads by inserting the missing elements.
Li, ______, K
How are elements grouped into various families in the periodic table?
The atomic number of an element 'X' is 11.
- Write the electronic configuration of X and find its valency.
- Write the formula and nature of its oxide.
Element 'X' forms an oxide with the formula X2O3 which is a solid with high melting point. ‘X’ would most likely be placed in the group of the Periodic Table as:
