Advertisements
Advertisements
प्रश्न
In which block of the modern periodic table are the nonmetals found?
पर्याय
s-block
p-block
d-block
f-block
Advertisements
उत्तर
In p-block of the modern periodic table are the nonmetals found.
APPEARS IN
संबंधित प्रश्न
An element P (atomic number 20) reacts with an element Q (atomic number 17) to form a compound. Answer the following questions giving reason:
Write the position of P and Q in the Modern Periodic Table and the molecular formula of the compound formed when P reacts with Q.
Rewrite the following statement after correction, if necessary:
Isotopes are the elements of the same group.
An element A has an atomic number of 6. Another element B has 17 electrons in its one neutral atom.
(a) In which groups of the periodic table would you expect to find these elements?
(b) what type of bond is formed between A and B?
(c) Suggest a formula of the compound formed between A and B.
Rearrange the columns 2 and 3 so as to match with the column 1.
| Column 1 | Column 2 | Column 3 |
| i. Triad ii. Octave iii. Atomic number iv. Period v. Nucleus vi. Electron |
a. Lightest and negatively charged particle in all the atoms b. Concentrated mass and positive charge c. Average of the first and the third atomic mass d. Properties of the eighth element similar to the first e. Positive charge on the nucleus f. Sequential change in molecular formulae |
1.Mendeleev
2. Thomson
3. Newlands
4. Rutherford
5. Dobereiner
. Moseley
|
The molecular formula of the chloride of element X is XCl. This compound is a solid having a high melting point. Which of the following elements be present in the same group as X.
Give a reason why elements need to be properly classified.
If the distance between two Cl atoms in a Cl2 molecule is 1.98Å, then the radius of the Cl atom is ______.
Among the given species A–, A+, and A, the smallest one in size is ______.
What property forms the basis of identification?
An element X from group 2 of the Periodic Table reacts with Y from group 17 to form a compound. Give the formula of the compound.
Which one of the following statements is not correct about the trends in the properties of the elements of a group on going down in a group?
Which of the following is a transition element?
Which one of the following does not increase while moving down the group of the periodic table?
Arrange the following elements in the order of their decreasing metallic character
Na, Si, Cl, Mg, Al
Identify and name the metals out of the following elements whose electronic configurations are given below.
- 2, 8, 2
- 2, 8, 1
- 2, 8, 7
- 2, 1
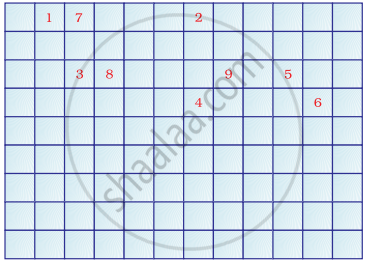
Complete the following cross word puzzle
Across:
(1) An element with atomic number 12.
(3) Metal used in making cans and member of Group 14.
(4) A lustrous non-metal which has 7 electrons in its outermost shell.
Down:
(2) Highly reactive and soft metal which imparts yellow colour when subjected to flame and is kept in kerosene.
(5) The first element of second Period
(6) An element which is used in making fluorescent bulbs and is second member of Group 18 in the Modern Periodic Table
(7) A radioactive element which is the last member of halogen family.
(8) Metal which is an important constituent of steel and forms rust when exposed to moist air.
(9) The first metalloid in Modern Periodic Table whose fibres are used in making bullet-proof vests
An element X of group 15 exists as diatomic molecule and combines with hydrogen at 773 K in presence of the catalyst to form a compound, ammonia which has a characteristic pungent smell.
- Identify the element X. How many valence electrons does it have?
- Draw the electron dot structure of the diatomic molecule of X. What type of bond is formed in it?
- Draw the electron dot structure for ammonia and what type of bond is formed in it?
Complete the following triads by inserting the missing elements.
Cl, ______, I
Complete the following table:
| Symbol | Atomic No | Electronic Configuration | |
| 1. | He | 2 | ______ |
| 2. | Ne | ______ | 2,8 |
| 3. | Ar | ______ | 2, 8,8 |
| 4. | Na | 11 | ______ |
