Advertisements
Advertisements
Question
Match the following
| 1. | Galvanisation | Noble gas elements |
| 2. | Calcination | Coating with Zn |
| 3. | Redox reaction | Silver-tin amalgam |
| 4. | Dental filling | Alumino thermic process |
| 5. | Group 18 elements | Heating in the absence of air |
Advertisements
Solution
| 1. | Galvanisation | Coating with Zn |
| 2. | Calcination | Heating in the absence of air |
| 3. | Redox reaction | Alumino thermic process |
| 4. | Dental filling | Silver-tin amalgam |
| 5. | Group 18 elements | Noble gas elements |
APPEARS IN
RELATED QUESTIONS
State whether the following statement is true of false:
The elements in a group have consecutive atomic numbers.
The non-metal A is an important constituent of our food and most of the fuels around us. A forms two oxides B and C. The oxide B is poisonous whereas oxide C causes global warming.
(a) Identify A, B and C.
(b) To which group of periodic table does A belong?
(c) Name another element which is placed in the same group as A.
A non-metal X which is the largest constituent of air combines with hydrogen when heated in the presence of iron as catalyst to form a gas Y. When gas Y is treated with sulphuric acid, it forms a compound Z which is used as a chemical fertiliser.
(a) What are X, Y and Z?
(b) To which group of periodic table does X belong?
(c) Name the periodic table in which X is placed.
(d) Which element is placed just before X in the period?
(e) Which element is placed just after X in the period?
Why was this basis of classification discarded for future classification of elements?
The basis of the classifications proposed by Dobereiner, Newlands and Mendeleev was ______.
Example for liquid metal is ______.
An element X combines with oxygen to form an oxide XO. This oxide is electrically conducting. Write the formula of the compound formed when X reacts with chlorine.
In which group are inert elements placed?
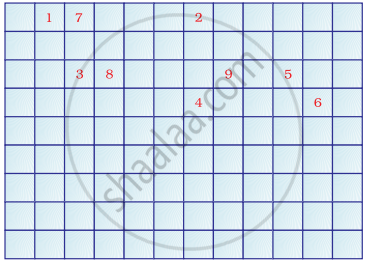
Complete the following cross word puzzle
Across:
(1) An element with atomic number 12.
(3) Metal used in making cans and member of Group 14.
(4) A lustrous non-metal which has 7 electrons in its outermost shell.
Down:
(2) Highly reactive and soft metal which imparts yellow colour when subjected to flame and is kept in kerosene.
(5) The first element of second Period
(6) An element which is used in making fluorescent bulbs and is second member of Group 18 in the Modern Periodic Table
(7) A radioactive element which is the last member of halogen family.
(8) Metal which is an important constituent of steel and forms rust when exposed to moist air.
(9) The first metalloid in Modern Periodic Table whose fibres are used in making bullet-proof vests
Complete the following triads by inserting the missing elements.
Li, ______, K
