Advertisements
Advertisements
Question
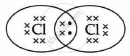
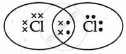
The electron dot structure of chlorine molecule is:
Options
Advertisements
Solution

Explanation:
Cl2 is the molecule of chlorine. Chlorine has an electronic configuration of 2, 8, and 7. Here, a combination of two chlorine atoms with 7 valence electrons each occurs by sharing 1 electron.
APPEARS IN
RELATED QUESTIONS
Name the type of bonds formed in ionic compounds and in the compounds formed by carbon.
Give one example of a molecule containing a double covalent bond
The solution of one of the following compounds will not conduct electricity. This compounds is:
(a) NaCl
(b) CCl4
(c) MgCl2
(d) CaCl2
The electronic configurations of two elements A and B are given below:
| A | 2, 6 |
| B | 2, 8, 1 |
(a) What type of chemical bond is formed between the two atoms of A?
(b) What type of chemical bond will be formed between the atoms of A and B?
For each compound mentioned above give the formulae of ions formed in aqueous solution.
Elements Q and S react together to form an ionic compound. Under normal conditions, which physical state will the compound QS exist in?
Taking MgCl2 as an electrovalent compound, CCl4 as a covalent compound, give four difference between electrovalent and covalent compounds
Write scientific reason.
Carbon has the property of forming many compounds.
Carbon can neither form C4- cation nor C4 anion. Why?
The number of single and double bonds present in a molecule of benzene (C6H6) respectively, are ______.