Advertisements
Advertisements
प्रश्न
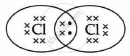
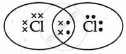
The electron dot structure of chlorine molecule is:
पर्याय
Advertisements
उत्तर

Explanation:
Cl2 is the molecule of chlorine. Chlorine has an electronic configuration of 2, 8, and 7. Here, a combination of two chlorine atoms with 7 valence electrons each occurs by sharing 1 electron.
APPEARS IN
संबंधित प्रश्न
Name a carbon containing molecule which has two double bonds.
Fill in the blank in the following sentence:
In forming N2 molecule, .............. electrons are shared by each atom of nitrogen.
A covalent molecule having a double bond between its atoms is:
(a) Hydrogen
(b) Oxygen
(c) water
(d) ammonia
One of the following contains a double bond as well as single bonds. This is:
(a) CO2
(b) O2
(c) C2H4
(d) C2H2
Explain the following term with example.
Hetero atom in a carbon compound
Taking hydrogen chloride and methane as examples, distinguish between a polar covalent bond and a non polar covalent bond.
Fill in the blank from the choice given in bracket.
The compound that does not have a lone pair of electrons is ___________. (Water, Ammonia, carbon tetrachloride)
Compound X consists of molecules.
Choose the letter corresponding to the correct answer from the options A, B, C and D given below:
X is likely to have a ______.
Complete the following activity.
Write the names of the hydrocarbons for the following structural formula.
(isobutylene, cyclohexane, propene, cyclohexene, cyclopentane, benzene, propyne, isobutane, propane)
 |
Oils on treating with hydrogen in the presence of palladium or nickel catalyst form fats. This is an example of