Advertisements
Advertisements
Question
The diagram given below is of an experiment just at the start. Study the diagram carefully and answer the following questions :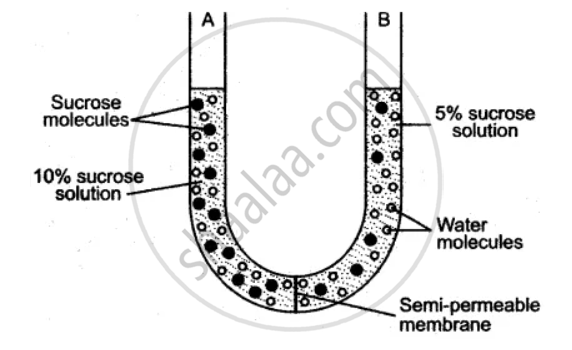
(i) What does the experiment demonstrate?
(ii) Define, the process demonstrated in the experiment.
(iii) What changes are observed after a few hours?
(iv) Give two examples of a semi-permeable membrane.
(v) Which limb of the U-tube contains a more concentrated sucrose solution, A or B?
(vi) Why is the membrane separating the two solutions labeled as semi-permeable membrane?
Advertisements
Solution
(i) The process of osmosis.
(ii) Osmosis is a special type of diffusion of solvent molecules through a semi-permeable membrane from a region of their higher concentration to the lower concentration region, but not vice versa.
(iii) The level of water in column A will rise along with the passage of time and will ultimately stop when the concentration of water molecules are equal on both sides of the membrane.
(iv) (a) Parchment paper, (b) Egg membrane.
(v) Solution A.
(vi) It permits the movement of only water molecules to pass through it.
APPEARS IN
RELATED QUESTIONS
Differentiate between Diffusion and Osmosis (Definition)
The highest water potential (capacity to move out higher concentrated solution) is that of ______.
Name the following:
The condition of a cell placed in a hypotonic solution.
Two potato cubes each 1 cm3 in size, were placed separately in two containers (A&B), the container (A) having water and the other (B) containing concentrated sugar solution. After 24 hours when the cubes were examined, those placed in water were found to be firm and had increased slightly in size and those placed in concentrated sugar solution were found to be soft and had somewhat decreased in size. Use the above information to answer the questions that follow:
Account for the firmness and increase in the size of the potato cubes placed in water.
What do you mean by transpiration pull ?
What is the significance of diffusion?
Give Reasons for the following.
Drops of water on a leaf of plant like peepal does not enter the leaf by osmosis?
Give Reasons for the following.
Plants growing in fertilized soil are often found to wilt if the soil is not adequately watered. Why?
Give Technical Term for the following.
The pressure which is responsible for the movement of water molecules across the cortical cells of the root.
Mention, if the following statement is True or False. If false rewrite the wrong statement in its correct form:
Osmosis is defined as the movement of water from a concentrated sugar solution to a dilute sugar solution.
