Advertisements
Advertisements
Question
Show how will you synthesize 1-phenylethanol from a suitable alkene.
Advertisements
Solution
Alkene is treated with H2O in the presence of acid (acid catalyzed hydrolysis) to give 1-phenylmethanol
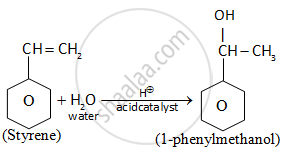
APPEARS IN
RELATED QUESTIONS
How will you convert: Propene to Propan-2-ol?
Name the reagents used in the following reactions:

Predict the major product of acid catalysed dehydration of butan-1-ol.
Write the mechanism of hydration of ethene to yield ethanol.
How is the following conversion carried out?
\[\ce{Benzyl chloride -> Benzyl alcohol}\]
How is the following conversion carried out?
\[\ce{Methyl magnesium bromide → 2-Methylpropan-2-ol}\]
Name the reagent used in the following reaction:
Dehydration of propan-2-ol to propene.

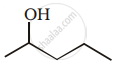
Show how you would synthesise the following alcohol from an appropriate alkene?
Write the structures of A, B and C in the following reactions :
The Wittig reaction is a reaction between a carbonyl compound (aldehyde or ketone only) and a species known as a phosphoniumylide. What is the expected final product in the Wittig reaction?
Carboxylic acids are more acidic than phenol and alcohol because of
A compound does not react with 2, 4 dinitrophenylhydrazine, the compound is:
The reagent used for dehydration of an alcohol is
When alcohol react with concentrated H2SO4 intermediate compound formed is
When glycol is heated with dicorboxylic acid the product are


The products "A" and "B" formed in above reactions are:
An aldehyde isomeric with allyl alcohol gives phenyl hydrazone. Pick out a ketone that too gives a phenyl hydrazone containing the same percentage of nitrogen.
The major product of the following reaction is:
\[\begin{array}{cc}
\ce{Cl}\phantom{.........................}\\
|\phantom{..........................}\\
\ce{CH3 - CH - CH3 ->[(i) Alc. KOH][(ii) HBr/peroxide (iii) aq. KOH]}
\end{array}\]
