Advertisements
Advertisements
Question
Predict the product of the following reaction:
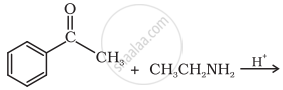
Advertisements
Solution
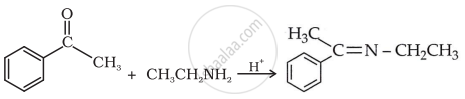
APPEARS IN
RELATED QUESTIONS
Write the product in the following reaction:

Write the structure of the product of the following reaction:

Write the reaction involved in the Stephen reduction
Ozonolysis of alkenes followed by the reaction with zinc dust and water gives ____________ depending on the substitution pattern of the alkene.
Esters react with DIBAL-H to produce:
Aldehydes are prepared by reducing nitriles to corresponding imines with stannous chloride in the presence of hydrochloric acid. This reaction is called:
Can Gatterman-Koch reaction be considered similar to Friedel Craft’s acylation? Discuss.
What is the name of the given reaction of preparation of aldehyde?
\[\ce{C3COCl ->[H2][Pd/BaSO4] CH3CHO + HCl}\]
\[\begin{array}{cc}
\ce{CH3 - CH2 - CH - CH3}\\
\phantom{.....}|\\
\phantom{......}\ce{Cl}
\end{array}\] obtained by chlorination of n-butane, will be
Benz aldehyde + NaOH →
The number of chiral carbon in glucose is:-
The intermediate compound ‘X’ in the following chemical reaction is:

The oxidation of toluene to benzaldehyde by chromyl chloride is called ______.
The reaction of benzene with CO and HCl in the presence of anhydrous AlCl3 gives ______.
Reagent used to convert allyl alcohol to acrolein is ______.
Assertion (A): Strong oxidising agents oxidise toluene and its derivatives to benzoic acids.
Reason (R): It is possible to stop the oxidation of toluene at the aldehyde stage with suitable reagents.
Select the most appropriate answer from the options given below:
Account for the following:
N-ethylbenzene sulphonyl amide is soluble in alkali.
Write the name of the reaction, structure and IUPAC name of the product formed when:
CH3CH2CN reacts with stannous chloride in the presence of hydrochloric acid, followed by hydrolysis.
