Advertisements
Advertisements
प्रश्न
Predict the product of the following reaction:
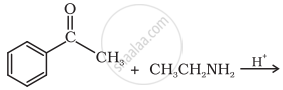
रासायनिक समीकरण/संरचनाएँ
Advertisements
उत्तर
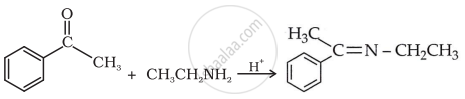
shaalaa.com
क्या इस प्रश्न या उत्तर में कोई त्रुटि है?
अध्याय 8: Aldehydes, Ketones and Carboxylic Acids - Intext Questions [पृष्ठ २४३]
