Advertisements
Advertisements
Question
Photocurrent recorded in the microammeter in an experimental setup of the photoelectric effect vanishes when the retarding potential is more than 0.8 V if the wavelength of incident radiation is 4950 Å. If the source of incident radiation is changed, the stopping potential turns out to be 1.2 V. Find the work function of the cathode material and the wavelength of the second source.
Advertisements
Solution
Data: VO = 0.8 V, λ = 4950 Å = 4.950 x 10-7 m, `"V"_"O"'` = 1.2 V, h = 6.63 × 10-34 J.s, c = 3 × 108 m/s
1. `"V"_"O""e" = "hv" - phi = "hc"/lambda - phi`
∴ The work function of the cathode material,
`phi = "hc"/lambda - "V"_"O""e"`
`= ((6.63 xx 10^-34)(3 xx 10^8))/(4.950 xx 10^-7) - (0.8)(1.6 xx 10^-19)`
= 4.018 × 10-19 - 1.28 × 10-19
= 2.738 × 10-19 J
`= (2.738 xx 10^-19 "J")/(1.6 xx 10^-19 "J"//"eV")`
= 1.711 eV
2. `"V"_"O"'"e" = "hc"/(lambda') - phi`
∴ `"hc"/(lambda') = "V"_"O"'"e" + phi`
∴ The wavelength of the second source,
`lambda' = "hc"/("V"_"O"'"e" + phi)`
`= ((6.63 xx 10^-34)(3 xx 10^8))/((1.2)(1.6 xx 10^-19) + 2.738 xx 10^-19)`
`= (19.89 xx 10^-26)/(4.658 xx 10^-19)`
= 4.270 × 10-7 m
= 4270 Å
APPEARS IN
RELATED QUESTIONS
If the frequency of incident light falling on a photosensitive material is doubled, then the kinetic energy of the emitted photoelectron will be ______.
Using the values of work function given in the following table, tell which metal will require the highest frequency of incident radiation to generate photocurrent.
Typical values of work function for some common metals
| Metal | Work function (in eV) |
| Potassium | 2.3 |
| Sodium | 2.4 |
| Calcium | 2.9 |
| Zinc | 3.6 |
| Silver | 4.3 |
| Aluminium | 4.3 |
| Tungsten | 4.5 |
| Copper | 4.7 |
| Nickel | 5.0 |
| Gold | 5.1 |
The electrons are emitted in the photoelectric effect from a metal surface.
Define photoelectric effect.
The minimum frequency for photoelectric effect on metal is 7 × 1014 Hz, Find the work function of the metal.
The work function of a surface is 3.1 eV. A photon of frequency 1 × 1015 Hz. Is an incident on it. Calculate the incident wavelength is photoelectric emission occurs or not.
The energy of the incident photon on the metal surface is 3 W and then 5 W, where W is the work function for that metal. The ratio of velocities of emitted photoelectrons is ______.
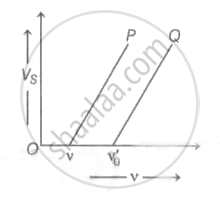
The graph of stopping potential `"V"_"s"` against frequency v of incident radiation is plotted for two different metals P and Q as shown in the graph. ΦP and ΦQ are work-functions of P and Q respectively, then
For photoelectric emission from certain metal, the cut-off frequency is v. If radiation of frequency 2v impinges on the metal plate, the maximum possible velocity of the emitted electron will be (m is the electron mass) ____________.
When a light of wavelength 4000 Å falls on a photoelectric emitter, photoelectrons are liberated. For another emitter, light of wavelength 6000 Å is sufficient for photo emission. The work functions of the two emitters are in the ratio of ____________.
The work function of a metallic surface is 5.01 eV. The photoelectrons are emitted when light of wavelength 2000 Å falls on it. The potential difference applied to stop the fastest photoelectrons is [h = 4.14 x 10-15 eV sec] ____________.
An important spectral emission line has a wavelength of 21 cm. The corresponding photon energy is (h = 6.62 x 10-34 Js, c = 3 x 108 m/s) ____________.
The photo electric effect to take place for a metal, the minimum frequency required is 5.792 × 1014 Hz. A light of wavelength 6000 Å is incident on that metal surface. What is the corresponding frequency of light and will there be photoelectric emissions? [velocity of light = 3 × 108 m/s]
The work function of a substance is 4.0 eV. The longest wavelength of light that can cause photo-emission from this substance is approximately (h = 6.63 × 10-34 Js)[1eV = 1.6 × 10-19 J]
The photon of frequency vis incident on a metal surface whose threshold frequency is v0. The kinetic energy of the emitted photoelectrons will be ____________.
The photon of frequency vis incident on a metal surface whose threshold frequency is v0. The kinetic energy of the emitted photoelectrons will be ______.
When a photosensitive surface is irradiated by lights of wavelengths `lambda_1` and `lambda_2`, kinetic energies of emitted photoelectrons are E1 and E2 respectively. The work function of the photosensitive surface is ____________.
An electromagnetic wave of wavelength '`lambda`' is incident on a photosensitive surface of negligible work function. If the photoelectrons emitted from this surface have the de-Broglie wavelength '`lambda_1`' then ____________.
A light of frequency 'v' is incident on the metal surface whose threshold frequency is 'v0'. If v = v0, then [c = speed of light in medium] ____________.
In a photoelectric experiment, ultraviolet light of wavelength 280 nm is used with a lithium cathode having work function Φ = 2.5 eV. If the wavelength of incident light is switched to 400 nm, find out the change in the stopping potential.
(h = 6.63 × 10-34 Js, c = 3 × 108 ms-1)
When radiation of wavelength λ is used to illuminate a metallic surface, the stopping potential is V. When the same surface is illuminated with radiation of wavelength 3λ, the stopping potential is `"V"/4`. If the threshold wavelength for the metallic surface is nλ. then value of n will be ______.
When ultraviolet light of wavelength 100 nm is incident upon a sample of silver metal, a potential difference of 7.7 volt is required to stop the photoelectrons from reaching the collector plate. The potential required to stop photo electrons when light of wavelength 200 nm is incident upon silver is ______.
For a given photosensitive material and frequency (> threshold frequency) of incident radiation, the photoelectric current varies with the intensity of incident light as:
In a photocell, frequency of incident radiation is increased by keeping other factors constant (v > v0), the stopping potential ______.
The photoelectric threshold for a certain metal surface is 3600 Å. If the metal surface is irradiated by a wavelength of 1100 Å, then kinetic energy of the emitted photoelectrons is ______.
Explain the failure of wave theory of light to account for the observations from experiments on photoelectric effect.
