Advertisements
Advertisements
Question
The threshold wavelength of tungsten is 2.76 x 10-5 cm.
(a) Explain why no photoelectrons are emitted when the wavelength is more than 2.76 x 10-5 cm.
(b) What will be the maximum kinetic energy of electrons ejected in each of the following cases
(i) if ultraviolet radiation of wavelength λ = 1.80 × 10-5 cm and
(ii) radiation of frequency 4 x 1015 Hz is made incident on the tungsten surface?
Advertisements
Solution
Data: λ0 = 2.76 x 10-5 cm = 2.76 x 10-7 m,
λ = 1.80 × 10-5 cm = 1.80 × 10-7 m,
v = 4 × 1015 Hz,
h = 6.63 × 10-34 J.s,
c = 3 × 108 m/s
(a) For λ > λ0,v < v0 (threshold frequency).
∴ hv < hv0. Hence, no photoelectrons are emitted.
(b) Maximum kinetic energy of electrons ejected
`= "hc" (1/lambda - 1/lambda_0)`
`= (6.63 xx 10^-34)(3 xx 10^8)(10^7/1.8 - 10^7/2.76)`J
= (6.63 × 10-19)(3)(0.5555 - 0.3623)
= (6.63)(3)(0.1932 × 10-19)J
= 3.842 × 10-19J
`= (3.842 xx 10^-19 "J")/(1.6 xx 10^-19 "J"//"eV")`
= 2.40 eV
(c) Maximum kinetic energy of electrons ejected
= hv - `"hc"/lambda_0`
`= (6.63 xx 10^-34)(4 xx 10^15) - ((6.63 xx 10^-34)(3 xx 10^8))/(2.76 xx 10^-7)`
= 26.52 × 10-19 - 7.207 × 10-19
= 19.313 × 10-19 J
= `(19.313 xx 10^19 "J")/(1.6 xx 10^-19 "J"//"eV")`
= 12.07 eV
RELATED QUESTIONS
If the frequency of incident light falling on a photosensitive material is doubled, then the kinetic energy of the emitted photoelectron will be ______.
Explain the inverse linear dependence of stopping potential on the incident wavelength in a photoelectric effect experiment.
Using the values of work function given in the following table, tell which metal will require the highest frequency of incident radiation to generate photocurrent.
Typical values of work function for some common metals
| Metal | Work function (in eV) |
| Potassium | 2.3 |
| Sodium | 2.4 |
| Calcium | 2.9 |
| Zinc | 3.6 |
| Silver | 4.3 |
| Aluminium | 4.3 |
| Tungsten | 4.5 |
| Copper | 4.7 |
| Nickel | 5.0 |
| Gold | 5.1 |
Photocurrent recorded in the microammeter in an experimental setup of the photoelectric effect vanishes when the retarding potential is more than 0.8 V if the wavelength of incident radiation is 4950 Å. If the source of incident radiation is changed, the stopping potential turns out to be 1.2 V. Find the work function of the cathode material and the wavelength of the second source.
Radiation of wavelength 4500 Å is incident on a metal having work function 2.0 eV. Due to the presence of a magnetic field B, the most energetic photoelectrons emitted in a direction perpendicular to the field move along a circular path of radius 20 cm. What is the value of the magnetic field B?
Given the following data for incident wavelength and the stopping potential obtained from an experiment on the photoelectric effect, estimate the value of Planck's constant and the work function of the cathode material. What is the threshold frequency and corresponding wavelength? What is the most likely metal used for emitter?
| Incident wavelength (in Å) | 2536 | 3650 |
| Stopping potential (in V) |
1.95 | 0.5 |
As the intensity of incident light increases ______
Find the energy of photon which have momentum 2 × 10-16 gm-cm/sec.
Draw a neat labelled diagram of a schematic of the experimental setup for the photoelectric effect.
The work function of a surface is 3.1 eV. A photon of frequency 1 × 1015 Hz. Is an incident on it. Calculate the incident wavelength is photoelectric emission occurs or not.
The energy of the incident photon on the metal surface is 3 W and then 5 W, where W is the work function for that metal. The ratio of velocities of emitted photoelectrons is ______.
When a photon enters glass from air, which one of the following quantity does not change?
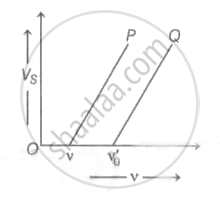
The graph of stopping potential `"V"_"s"` against frequency v of incident radiation is plotted for two different metals P and Q as shown in the graph. ΦP and ΦQ are work-functions of P and Q respectively, then
If the maximum kinetic energy of emitted electrons in photoelectric effect is 3.2 × 10-19 J and the work-function for metal is 6.63 × 10-19 J, then stopping potential and threshold wavelength respectively are
[Planck's constant, h = 6.63 × 1034 J-s]
[Velocity of light, c = 3 × 108 `"m"/"s"`]
[Charge on electron= 1.6 × 10-19 C]
Threshold wavelength for lithium metal is 6250 Å. For photoemission, the wavelength of the incident light must be ______.
In photoelectric effect, for a light of different intensities but of same frequency, the stopping potential for a given metal is ____________.
In photoelectric effect, graph of saturation current versus frequency of light is plotted. The nature of the graph will be ____________.
When wavelength of incident radiation on the metal surface is reduced from 'λ1' to 'λ2', the kinetic energy of emitted photoelectrons is tripled. The work function of the metal is ______.
(h = Planck's constant, c =velocity of light)
The photon of frequency vis incident on a metal surface whose threshold frequency is v0. The kinetic energy of the emitted photoelectrons will be ______.
The work function of a photosensitive material is 4.0 eV. The longest wavelength of light that can cause photon emission from the substance is (approximately) ____________.
Which one of the following statements ts INCORRECT for stopping potential in photoelectric emission?
A light of wavelength '`lambda`' and intensity 'I' falls on photosensitive material. If 'N' photoelectrons are emitted, each with kinetic energy E, then ____________.
Two incident radiations having energies two times and ten times of the work function of a metal surface, produce photoelectric effect. The ratio of maximum velocities of emitted photo electrons respectively is ____________.
The radiation corresponding to the 3 → 2 transition of a hydrogen atom falls on a gold surface to generate photoelectrons. These electrons are passed through a magnetic field of 5 × 10-4 T. Assume that the radius of the largest circular path followed by these electrons is 7 mm, and the work function of the metal is ______.
(Mass of electron = 9.1 × 10-31 kg)
The stopping potential in the context of photoelectric effect depends on the following property of incident electromagnetic radiation ______.
The wavelength of light incident on a metal surface is reduced from 300 nm to 200 nm (both are less than threshold wavelength). What is the change in the stopping potential for photoelectrons emitted from the surface will be ______ V. (Take h = 6.6 × 10-34 J-s)
For a given photosensitive material and frequency (> threshold frequency) of incident radiation, the photoelectric current varies with the intensity of incident light as:
Define photoelectric work function of a metal.
Give Einstein's explanation of the photoelectric effect.
