Advertisements
Advertisements
Question
Observe the structural formula and answer the following questions.
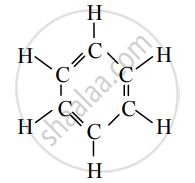
- Write the name of the given hydrocarbon.
- The given hydrocarbon is included in which type of hydrocarbon?
- What is the kind of compounds with the above characteristic structure called?
Advertisements
Solution
- The given hydrocarbon is benzene.
- The given hydrocarbon is included in cyclic hydrocarbon.
- The compounds having this characteristic unit in their structure are called aromatic compounds.
APPEARS IN
RELATED QUESTIONS
Fill in the following blank with suitable word:
Ethene and ethyne are examples of ..... hydrocarbons.
By how many carbon atoms and hydrogen atoms do any two adjacent homologues differ?
Define a homologous series. Give the name and structural formula of one homologue of the following:
CH3OH
Propane and ethane are ______.
What is the difference in the molecular formula of any two adjacent homologues in terms of molecular mass?
 .
.
Write names of first four homologous series of alcohols.

What is the difference in the molecular formula of any two adjacent homologues:
(i) In terms of molecular mass
(ii) In terms of number and kind of atoms per molecule?
The general formula of Alkane is _________________
Assertion (A): In a homologous series of alcohols, the formula for the second member is C2H5OH and the third member is C3H7OH.
Reason (R): The difference between the molecular masses of the two consecutive members of a homologous series is 144.
Saturated hydrocarbon : Single bond : : Unsaturated hydrocarbon : _______
What is called homologous series? Give any three of its characteristics?
Which of the following does not belong to the same homologous series?
A carbon compound ‘A’ having melting point 156K and boiling point 351K, with molecular formula C2H6O is soluble in water in all proportions.
- Identify ‘A’ and draw its electron dot structure.
- Give the molecular formulae of any two homologues of ‘A’.
Consider the carbon compounds having following molecular formula:
(i) C3H6 (ii) C3H8 (iii) C4H6 (iv) C6H6 (v) C6H12
- State the number of double covalent bonds present in C3H8.
- Write the formula of first member of the homologous series to which the carbon compound C4H6 belongs.
- Which one of the above compounds forms a ring structure of carbon atoms?
- Identify, which of the above compounds, is a member of alkane series.
Name and draw the electron dot structure of first homologue of alkynes series.
