Advertisements
Advertisements
Question
Mercury has an angle of contact equal to 140° with soda lime glass. A narrow tube of radius 1.00 mm made of this glass is dipped in a trough containing mercury. By what amount does the mercury dip down in the tube relative to the liquid surface outside? Surface tension of mercury at the temperature of the experiment is 0.465 N m–1. Density of mercury = 13.6 × 103 kg m–3
Advertisements
Solution 1
Angle of contact between mercury and soda lime glass, θ = 140°
Radius of the narrow tube, r = 1 mm = 1 × 10–3 m
Surface tension of mercury at the given temperature, s = 0.465 N m–1
Density of mercury, ρ =13.6 × 103 kg/m3
Dip in the height of mercury = h
Acceleration due to gravity, g = 9.8 m/s2
Surface tension is related with the angle of contact and the dip in the height as:
`s = (hrhogr)/(2costheta)`
`:.h = (2s cos theta)/(rrhog)`
` = (2xx0.465xxcos 140)/(1xx10^(-3)xx13.6xx10^3xx 9.8)`
= - 0.00534 m
= -5.34 mm
Here, the negative sign shows the decreasing level of mercury. Hence, the mercury level dips by 5.34 mm
Solution 2
Radius or tube = r = 1.00 mm = `10^(-3) m`
Surface tension of mercury, `sigma = 0.465 Nm^(-1)`
Density of mercury, `sigma = 13.6 xx 10^3 kg m6(-3)`
Angle of contact, `theta = 140^@`
`:. h = (2sigma cos theta)/(rrhog) = (2xx0.465xxcos 140^@)/(10^(-3)xx 13.6xx10^3xx 9.8)`
`= (2xx0.465xx(-0.7660))/(10^(-3)xx13.6xx10^3xx9.8)`
`= -5.34 xx 10^(-3) m = -5.34 mm`
Negative sign shows that the mercury level is deprssedin the tube
RELATED QUESTIONS
Angle of contact for the pair of pure water with clean glass is _______.
Explain why Surface tension of a liquid is independent of the area of the surface
If water in one flask and castor oil in other are violently shaken and kept on a table, which will come to rest earlier?
A heavy mass is attached to a thin wire and is whirled in a vertical circle. The wire is most likely to break
The excess pressure inside a soap bubble is twice the excess pressure inside a second soap bubble. The volume of the first bubble is n times the volume of the second where n is
The contact angle between a solid and a liquid is a property of
(a) the material of the solid
(b) the material of the liquid
(c) the shape of the solid
(d) the mass of the solid
Find the excess pressure inside (a) a drop of mercury of radius 2 mm (b) a soap bubble of radius 4 mm and (c) an air bubble of radius 4 mm formed inside a tank of water. Surface tension of mercury, soap solution and water are 0.465 N m−1, 0.03 N m−1 and 0.076 N m−1 respectively.
A capillary tube of radius 1 mm is kept vertical with the lower end in water. (a) Find the height of water raised in the capillary. (b) If the length of the capillary tube is half the answer of part , find the angle θ made by the water surface in the capillary with the wall.
Explain the capillary action.
Twenty-seven droplets of water, each of radius 0.1 mm coalesce into a single drop. Find the change in surface energy. Surface tension of water is 0.072 N/m.
The property of _______ of a liquid surface enables the water droplets to move upward in plants.
Define the angle of contact for a given pair of solid and liquid.
What is capillarity?
Soap solution is used for cleaning dirty clothes because ______.
What is surface tension? Explain the applications of surface tension.
A hot air balloon is a sphere of radius 8 m. The air inside is at a temperature of 60°C. How large a mass can the balloon lift when the outside temperature is 20°C? (Assume air is an ideal gas, R = 8.314 J mole–1K–1, 1 atm. = 1.013 × 105 Pa; the membrane tension is 5 Nm–1.)
A soap bubble of radius 3 cm is formed inside another soap bubble of radius 6 cm. The radius of an equivalent soap bubble which has the same excess pressure as inside the smaller bubble with respect to the atmospheric pressure is ______ cm.
A drop of water and a soap bubble have the same radii. Surface tension of soap solution is half of that of water. The ratio of excess pressure inside the drop and bubble is ______.
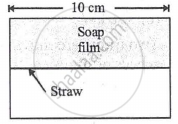
A soap film of surface tension 3 × 10-2 formed in a rectangular frame can support a straw as shown in Fig. If g = 10 ms-12, the mass of the straw is ______.
A spherical liquid drop of radius R is divided into eight equal droplets. If surface tension is T, then the work done in this process will be ______.
